At a glance
- Phase 2 obesity: 14.9% mean weight loss at 4.8 mg over 46 weeks (Le Roux et al. Lancet D&E 2024)
- Phase 2 MASH: 62% reached histologic improvement at 4.8 mg vs 14% on placebo (Sanyal et al. NEJM 2024)
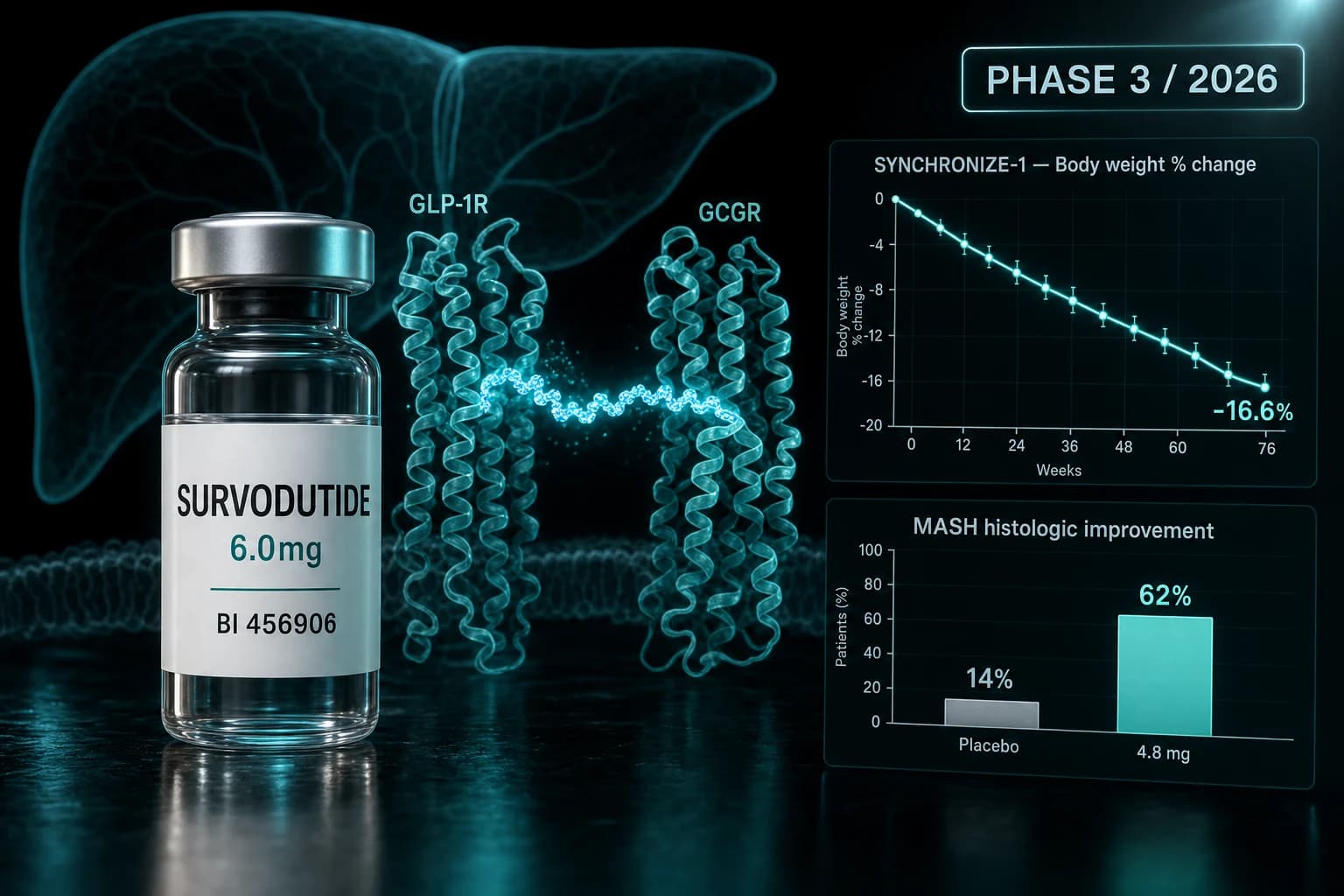
- SYNCHRONIZE-1 Phase 3 topline: 16.6% mean weight loss at 76 weeks vs 3.2% on placebo
- Glucagon receptor adds hepatic fat oxidation and energy expenditure on top of GLP-1 satiety
- GI side effects in roughly 75% of recipients; serious AEs lower than placebo in Phase 2
- Not FDA-approved; full Phase 3 data due in 2026 peer-reviewed publication and ADA Scientific Sessions
Survodutide is the dual agonist closing the gap to triple agonist weight loss
Boehringer Ingelheim and Zealand Pharma's survodutide (BI 456906) is a once-weekly subcutaneous peptide that activates two receptors at once: GLP-1R for appetite suppression and the glucagon receptor (GCGR) for hepatic fat oxidation and energy expenditure. In April 2026, SYNCHRONIZE-1 reported 16.6% mean weight loss at 76 weeks in adults with obesity but no diabetes, against 3.2% on placebo. That puts survodutide between tirzepatide and retatrutide on the obesity efficacy spectrum, while opening a separate clinical door on liver disease.
The Phase 2 program already published two readouts that matter. In a 387-patient dose-finding trial, the 4.8 mg dose produced 14.9% mean weight loss at 46 weeks (Le Roux et al. Lancet D&E 2024). In a separate 295-patient liver trial, 62% of participants on 4.8 mg achieved improvement in MASH with no worsening of fibrosis at 48 weeks, versus 14% on placebo (Sanyal et al. NEJM 2024).
This is what the published evidence actually shows on survodutide, what the GCG/GLP-1 mechanism adds beyond tirzepatide and semaglutide, and where it fits against retatrutide for researchers tracking the class through 2026.
What survodutide is at the molecule level
Survodutide is a 29-amino-acid linear peptide with C-terminal lipidation, designed for once-weekly dosing. The receptor profile is what separates it from semaglutide and tirzepatide. Unlike semaglutide (GLP-1R only) or tirzepatide (GLP-1R + GIP-R), survodutide pairs GLP-1R with GCGR, the same axis Lilly later extended to a triple agonist by adding GIP-R activity.
Glucagon receptor agonism is not a new idea in metabolic pharmacology. The proof of concept goes back to a single-molecule chimera of glucagon and exendin-4 that produced sustained weight loss in diet-induced obese mice (Day et al. Nat Chem Biol 2009). What was missing for fifteen years was a tolerable, once-weekly clinical molecule. Survodutide is the first GCG/GLP-1 dual to publish Phase 2 readouts in two indications and to advance into a multi-arm Phase 3 program.
The Phase 3 program is called SYNCHRONIZE. It includes:
- SYNCHRONIZE-1 in adults with obesity, no type 2 diabetes
- SYNCHRONIZE-2 in adults with obesity and type 2 diabetes (Wharton et al. DOM 2026)
- A cardiovascular outcomes trial powered for MACE
- A separate Phase 3 liver program in MASH with fibrosis
The dosing strategy in trials is once-weekly subcutaneous injection with a slow up-titration to either 3.6 mg or 6.0 mg. Slow titration matters here, because GCGR agonism brings its own GI burden on top of GLP-1.
Why glucagon receptor agonism matters for weight loss
GLP-1R agonism reduces body weight primarily through appetite suppression: it slows gastric emptying and acts on hypothalamic and brainstem circuits to lower food intake. That mechanism has a ceiling. At maximum tolerated GLP-1 doses, the additional gain from pushing further is mostly noise.
Glucagon receptor agonism contributes through a different lever. In hepatocytes, GCGR activation increases fatty acid oxidation and ketogenesis. In adipose tissue and the liver as a whole, it raises energy expenditure. The translation into rodent models is well-characterized: dual GLP-1/glucagon co-agonism reverses obesity in mice with greater fat-mass loss than GLP-1 alone, even at matched food intake (Day et al. 2009).
The clinical question has always been whether you can dial in enough GCGR signal to get the energy expenditure benefit without unmasking the diabetogenic side of glucagon. Pure glucagon raises hepatic glucose output. Survodutide's dose ratio is tuned so that GLP-1R-mediated insulinotropic effects offset the hepatic glucose contribution from GCGR. SYNCHRONIZE-2 will be the cleanest test of that balance in people with type 2 diabetes.
Bottom line: Survodutide adds liver-side fat oxidation to GLP-1 satiety. The mechanism is why obesity efficacy can keep climbing past the GLP-1-only ceiling, and why the same molecule has a real shot at MASH.
Phase 2 obesity: 14.9% weight loss at 46 weeks
The pivotal Phase 2 dose-finding trial enrolled 387 adults with BMI of at least 27 and no type 2 diabetes, randomizing them across four survodutide doses and placebo for 46 weeks (Le Roux et al. Lancet D&E 2024).
| Group | n | Mean weight change at 46 weeks |
|---|---|---|
| Placebo | 77 | -2.8% |
| Survodutide 0.6 mg | 77 | -6.2% |
| Survodutide 2.4 mg | 78 | -12.5% |
| Survodutide 3.6 mg | 77 | -13.2% |
| Survodutide 4.8 mg | 78 | -14.9% |
A few details matter for interpretation:
- 55% of participants on 4.8 mg reached at least 15% body weight loss
- The weight curve had not flattened at 46 weeks, suggesting the on-treatment plateau lies further out
- Discontinuations for adverse events were higher on survodutide than placebo, but serious adverse event rates were lower (4.2% vs 6.5%)
The 14.9% figure is the on-treatment estimand, which counts data while participants stay on drug. The trial-product estimand (intent-to-treat with treatment-policy adjustments) ran a few points lower, in line with how every modern obesity trial reports two estimands now.
A post-hoc analysis from the same trial showed mean systolic blood pressure dropped 8.6 mmHg on 4.8 mg versus 2.5 mmHg on placebo at 46 weeks, with a 4.8 mmHg diastolic reduction (Le Roux et al. DOM 2025). Blood pressure changes track weight loss across this class, but the magnitude here is consistent with what tirzepatide and semaglutide produce at comparable percentage reductions.
Phase 2 MASH: histologic improvement at 48 weeks
The MASH readout was published two months after the obesity trial, in NEJM (Sanyal et al. 2024). The design used liver biopsy as the primary endpoint, which is the regulatory gold standard for MASH drug development.
Patient population:
- 295 adults with biopsy-confirmed MASH and fibrosis stage F1 to F3
- Once-weekly survodutide at 2.4 mg, 4.8 mg, or 6.0 mg, or placebo
- 48 weeks of treatment with end-of-treatment biopsy
Primary endpoint (improvement in MASH with no worsening of fibrosis):
| Group | Histologic responders |
|---|---|
| Placebo | 14% |
| Survodutide 2.4 mg | 47% |
| Survodutide 4.8 mg | 62% |
| Survodutide 6.0 mg | 43% |
The dose-response is not strictly monotonic. The 6.0 mg arm underperformed 4.8 mg, which the investigators attributed in part to higher discontinuation rates at the top dose. That pattern is recurring across this class: the highest tolerated dose is not always the highest pharmacologic dose.
Secondary endpoints included improvement in fibrosis without worsening of MASH, achieved by 36% on 4.8 mg versus 22% on placebo. Liver fat reduction by MRI-PDFF was substantial across all active arms, in the range of 60% relative reduction at the higher doses.
Note: A 62% histologic response rate at 4.8 mg is competitive with resmetirom, the FDA-approved THR-beta agonist. Survodutide is now in Phase 3 for MASH with fibrosis, separate from the obesity program.
SYNCHRONIZE-1: the Phase 3 obesity topline
In April 2026, Boehringer Ingelheim and Zealand Pharma reported topline results from SYNCHRONIZE-1, the pivotal Phase 3 obesity trial in adults without type 2 diabetes. The trial enrolled participants with BMI of at least 30, or 27 with weight-related comorbidities, and ran for 76 weeks.
Headline numbers from the topline release:
- Mean weight loss of 16.6% at 76 weeks (efficacy estimand) vs 3.2% on placebo
- 85.1% of participants on survodutide achieved at least 5% reduction
- Both co-primary endpoints met
- Full peer-reviewed data planned for the American Diabetes Association Scientific Sessions in June and a journal publication later in 2026
The 16.6% number is the on-treatment efficacy estimand, the same convention SURMOUNT-1 used to report tirzepatide's 22.5% at 72 weeks and STEP 1 used for semaglutide's 14.9% at 68 weeks. Compared head-to-head on the same estimand, survodutide is below tirzepatide and slightly above semaglutide, while approaching but not matching retatrutide's Phase 2 numbers (retatrutide vs tirzepatide vs semaglutide).
Treat the 16.6% as preliminary until the full publication lands. Topline press releases give the headline endpoint and the safety summary, not the discontinuation curves, the responder distributions, or the secondary outcomes that determine how a drug actually performs in clinic.
SYNCHRONIZE-2: the diabetes arm
SYNCHRONIZE-2 is the parallel Phase 3 trial in adults with obesity and type 2 diabetes. Baseline characteristics for the 752 treated participants were published in late 2025 (Wharton et al. DOM 2026).
What to expect from the readout:
- Two active arms (3.6 mg and 6.0 mg) plus placebo, randomized 1:1:1
- 76 weeks of treatment
- Co-primary endpoints: percent change in body weight and proportion achieving at least 5% loss
- Key secondary: change in HbA1c, given the diabetic population
SYNCHRONIZE-2 stress-tests the GCG/GLP-1 dose balance in a diabetic population. In Phase 2 of the obesity program, survodutide produced HbA1c-lowering effects comparable to other incretin-based therapies in non-diabetic participants. The Phase 3 readout will determine whether glucagon receptor agonism creates problems in glycemic control when the pancreas is already compromised, or whether the GLP-1R contribution carries through.
Side effects, tolerability, and what discontinuations look like
Across the Phase 2 obesity trial, 91% of survodutide recipients had at least one adverse event, versus 75% on placebo. The dominant signal was gastrointestinal: nausea, vomiting, diarrhea, and constipation, in 75% of survodutide recipients versus 42% on placebo.
A few specifics:
- Most GI events occurred during dose escalation, not maintenance
- Discontinuations due to adverse events were higher on survodutide, but serious adverse events were rarer than on placebo (4.2% vs 6.5%)
- No new pancreatitis signals beyond what is already documented for GLP-1 receptor agonists
- Heart rate increased modestly on drug, in line with class effect
Two safety domains specific to GCGR co-agonism are worth tracking:
- Hepatic glucose output: Pure glucagon raises hepatic glucose production. In Phase 2 trials, survodutide did not worsen glycemia, but SYNCHRONIZE-2 will be the definitive test in people with type 2 diabetes.
- Lean mass and resting energy expenditure: GCGR agonism may increase resting energy expenditure, which in theory could affect lean mass loss patterns. The full GLP-1 class has a lean-mass loss signal of roughly 25 to 40% of total weight reduction; whether dual agonism shifts that ratio either way is still open.
Warning: Discontinuations from GI events were higher on survodutide than placebo. Researchers tracking the class should treat the slow titration schedule as load-bearing, not optional. Aggressive titration is the most common cause of discontinuation in GLP-1 and dual-agonist programs.
How survodutide compares to the rest of the class
Comparing across trials is imperfect, because trial duration, baseline BMI, and titration schedules differ. The table below uses the on-treatment efficacy estimand for each pivotal trial in adults with obesity and no diabetes, where reported.
| Compound | Receptor profile | Pivotal trial | Duration | Mean weight loss |
|---|---|---|---|---|
| Semaglutide 2.4 mg | GLP-1R | STEP 1 | 68 wk | 14.9% |
| Tirzepatide 15 mg | GLP-1R + GIP-R | SURMOUNT-1 | 72 wk | 22.5% |
| Retatrutide 12 mg | GLP-1R + GIP-R + GCGR | Phase 2 | 48 wk | 24.2% |
| Survodutide 6.0 mg | GLP-1R + GCGR | SYNCHRONIZE-1 | 76 wk | 16.6% (topline) |
| Cagrilintide + semaglutide 2.4 mg | Amylin + GLP-1R | REDEFINE 1 | 68 wk | 22.7% |
| Orforglipron 36 mg (oral) | GLP-1R | ATTAIN-1 | 72 wk | 12.4% |
A few honest reads of this table:
- Tirzepatide remains the head-to-head benchmark for any new injectable. Survodutide does not exceed it in obesity.
- Retatrutide's edge over survodutide likely comes from adding GIP-R activity to the GLP-1 + GCGR scaffold.
- Survodutide differentiates on liver, where it has a randomized biopsy-endpoint trial in MASH that no other dual or triple agonist has yet matched at this stage of development.
- For oral access, orforglipron wins on convenience while delivering less weight loss; survodutide stays in the injectable lane.
For context on the full incretin class, the retatrutide vs tirzepatide vs semaglutide head-to-head and the CagriSema REDEFINE Phase 3 guide are the closest reference points.
Open questions the Phase 3 program needs to answer
A few items remain unsettled until the full SYNCHRONIZE data and the MASH Phase 3 readout publish:
- Plateau timing. Phase 2 weight curves had not flattened at 46 weeks. If the curve plateaus around 18 to 20% on the 6.0 mg dose at 76 weeks, survodutide closes part of the gap to tirzepatide. If it plateaus at 16 to 17%, it stays a clear step below.
- MASH durability. Phase 2 MASH used a 48-week biopsy. Phase 3 MASH outcomes need to confirm fibrosis improvement at longer follow-up, with hard liver outcomes (cirrhosis progression, decompensation) as the eventual goal.
- Cardiovascular outcomes. SYNCHRONIZE CVOT runs in parallel. Whether GCGR co-agonism contributes additive cardiovascular benefit beyond GLP-1 on a class-effect basis is unknown.
- Maintenance dose. No public protocol yet covers what happens after the 76-week induction phase. Whether researchers can titrate down to maintain weight loss, or whether stopping triggers the same regain pattern documented for semaglutide and tirzepatide, will matter for long-term use.
- Lean mass and physical function. Body composition data have not been broken out yet for the Phase 3 program. Given the energy-expenditure mechanism, this is one of the more interesting open questions.
What this means for researchers tracking the class
Survodutide is a credible second-tier injectable in obesity and a possible best-in-class agent for MASH, depending on Phase 3 liver outcomes. It is not approved by the FDA, EMA, or any other regulator for any indication. None of the doses described here are commercially available outside of clinical trials. Publicly distributed reference standards exist for research use only and are not interchangeable with investigational drug product.
For practical comparisons against compounds that are accessible through compounded or research-grade channels today, the tirzepatide vs semaglutide head-to-head and the retatrutide explained deep-dive cover the real options the community is currently studying. For dosing logistics on injectable peptides generally, the reconstitution calculator handles the math without rounding errors.
If you research compounded incretin analogues, both Ascension Peptides (injectables, code ENHANCED for 50% off) and Limitless Biotech (orals, code ENHANCED) carry the approved-class compounds; survodutide itself is not yet available outside of trials and we will not stock or recommend any source claiming otherwise.
The next inflection point is the ADA Scientific Sessions in June 2026, where the full SYNCHRONIZE-1 data should drop. After that, watch for the SYNCHRONIZE-2 readout in T2D and the MASH Phase 3 results, both of which will determine whether survodutide carves out a real lane or stays a runner-up to tirzepatide and retatrutide.
This article is for educational and research purposes only. Survodutide is not FDA-approved for any indication and is investigational outside of clinical trials. Doses, durations, and route descriptions reflect what is reported in the cited peer-reviewed literature and topline announcements; they are not protocols for human use. Nothing here is medical advice. Consult a qualified clinician for individual medical questions.