At a glance
- SUMMIT cut cardiovascular death or worsening HF events 38% vs placebo (HR 0.62, 95% CI 0.41-0.95; Packer et al. NEJM 2024, PMID 39555826)
- KCCQ-CSS rose 6.9 points more than placebo at 52 weeks; 6-minute walk distance improved 18.3 m
- CMR substudy: LV mass fell 11 g and paracardiac adipose 45 mL vs placebo (Kramer et al. JACC 2024, PMID 39566869)
- Body weight fell about 12% on tirzepatide; greater weight loss tracked with larger gains in 6MWD and KCCQ-CSS
- Cardiovascular benefit held in patients with chronic kidney disease (Butler et al. JACC 2025, PMID 40162940)
- Trial enrolled obese HFpEF only (BMI 30+); the result does not generalize to lean HFpEF
Why SUMMIT mattered
Heart failure with preserved ejection fraction is the version of heart failure that has resisted almost every drug class. SGLT2 inhibitors finally cracked it. Then a GLP-1/GIP co-agonist showed up to a Phase 3 trial designed for hard cardiovascular endpoints and walked out with a 38% relative risk reduction.
That is the SUMMIT trial in one sentence. The full read is more interesting, because the result was not just driven by weight loss, the secondary endpoints reshaped how cardiologists are thinking about obesity-related HFpEF, and the data shipped with mechanistic substudies that explain why the drug worked.
This article is the structured Phase 3 read. For the broader tirzepatide context, the tirzepatide compound page covers pharmacology and the full SURMOUNT family. For the head-to-head against the GLP-1 monotherapies, the tirzepatide vs semaglutide 2026 head-to-head covers the broader weight-loss comparison. For the parallel non-weight-loss indication, the tirzepatide for sleep apnea read on SURMOUNT-OSA covers the FDA-approved sleep-apnea label.
What SUMMIT tested
SUMMIT was an international, double-blind, randomized, placebo-controlled Phase 3 trial enrolling 731 patients with NYHA class II to IV heart failure, an ejection fraction of at least 50%, and a body mass index of at least 30 kg/m^2 (Packer et al. NEJM 2024, PMID 39555826). Participants were randomized 1:1 to subcutaneous tirzepatide titrated to a maximum tolerated dose of 15 mg weekly or matching placebo, on top of background heart failure therapy.
Two primary endpoints were prespecified:
- Adjudicated cardiovascular death or a worsening heart failure event, analyzed as time-to-first-event over a median follow-up of 104 weeks.
- Change in the Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS) from baseline to 52 weeks. KCCQ-CSS is a patient-reported measure of heart-failure symptom burden and physical limitation. A 5-point change is generally considered clinically meaningful.
Secondary endpoints included 6-minute walk distance, body weight, hsCRP, systolic blood pressure, NT-proBNP, EQ-5D-5L quality of life, and a composite quality-of-life score. The two-primary structure mattered: the trial had to demonstrate both a hard outcome and a patient-reported outcome to qualify as a positive read.
Note: SUMMIT specifically required a BMI at or above 30 kg/m^2 and a recent history of either elevated NT-proBNP or structural heart disease consistent with HFpEF. The cohort skewed older (mean about 65), majority female, with high prevalence of atrial fibrillation, hypertension, and chronic kidney disease.
Headline results at 104 weeks
The composite primary endpoint of cardiovascular death or worsening heart failure occurred in 36 of 364 tirzepatide patients (9.9%) versus 56 of 367 placebo patients (15.3%). The hazard ratio was 0.62 (95% CI 0.41 to 0.95), translating to a 38% relative risk reduction over a median 104 weeks of follow-up.
| Endpoint | Tirzepatide | Placebo | Effect |
|---|---|---|---|
| CV death or worsening HF event | 9.9% (36/364) | 15.3% (56/367) | HR 0.62 (0.41-0.95) |
| Worsening HF events alone | 8.0% (29/364) | 14.2% (52/367) | HR 0.54 (0.34-0.85) |
| KCCQ-CSS change at 52 weeks | +19.5 points | +12.7 points | +6.9 (3.3-10.6) |
| 6-minute walk distance at 52 weeks | gain | smaller gain | +18.3 m |
| Body weight at 52 weeks | -13.9% | -2.2% | -11.6 percentage points |
| hsCRP at 52 weeks | substantial drop | small drop | -34.9% |
| Systolic blood pressure at 52 weeks | drop | small drop | about -5 mm Hg |
Numbers are drawn from the primary publication (PMID 39555826) and the Circulation clinical-trajectory secondary analysis (Zile et al. 2024, PMID 39556714), which reported hazard ratios of 0.41 to 0.67 across alternative composites of death and worsening heart failure events, with consistent direction across every prespecified subgroup analyzed.
Two things to flag in this table. The KCCQ-CSS effect of 6.9 points exceeds the standard 5-point threshold for clinical meaningfulness, and the 6MWD gain of 18.3 m is in the range typically considered patient-relevant for HFpEF trials. The trial cleared both primary endpoints with margin, not narrowly.
Bottom line: SUMMIT is the first Phase 3 trial in HFpEF where a non-SGLT2 pharmacologic agent reduced both hard cardiovascular events and patient-reported symptom burden in the same cohort. The result establishes a class effect of incretin-based therapy in obesity-related HFpEF, building on STEP-HFpEF.
Was it just weight loss
This is the question every HFpEF cardiologist asked the morning after the AHA 2024 presentation. The SUMMIT investigators were ready for it.
The Nature Medicine secondary analysis (Borlaug et al. 2024, PMID 39551891) tested whether tirzepatide reduced the markers of circulatory overload that define HFpEF physiology. Tirzepatide produced a placebo-adjusted reduction in systolic blood pressure of about 5 mm Hg, a decrease in estimated blood volume, a drop in NT-proBNP, and a roughly 35% drop in hsCRP at 52 weeks. The authors framed these as evidence that tirzepatide is unloading the cardiovascular system, not just shrinking the patient.
The SUMMIT cardiac MRI substudy (Kramer et al. JACC 2024, PMID 39566869) went further. In 106 patients who completed paired CMR imaging at baseline and 52 weeks, tirzepatide reduced left ventricular mass by 11 g (95% CI -19 to -4, P = 0.004) versus placebo and reduced paracardiac adipose tissue by 45 mL (95% CI -69 to -22, P less than 0.001). Both reductions correlated with body weight change but only partially: the LV mass effect tracked weight loss but also tracked waist-circumference and blood-pressure changes.
Read together, the mechanistic story includes weight loss as one variable but is broader. Tirzepatide reduced ectopic and visceral adiposity (paracardiac fat fell 45 mL), lowered systemic inflammation (hsCRP fell about 35%), dropped blood pressure and estimated intravascular volume, and structurally remodeled the obese heart (LV mass fell 11 g). These changes track weight change but only partially.
The BMI and weight-loss subanalysis (PMID 40701669) reinforced this: greater weight loss at 52 weeks correlated with larger improvements in 6MWD, KCCQ-CSS, hsCRP, and blood pressure, but the cardiovascular event benefit was preserved across BMI and weight-loss strata. Patients who lost less weight still benefited; patients who lost more weight benefited more on the symptom side.
SUMMIT vs STEP-HFpEF: tirzepatide vs semaglutide
Semaglutide got there first. The STEP-HFpEF trial (Kosiborod et al. NEJM 2023, PMID 37622681) randomized 529 patients with HFpEF and obesity to semaglutide 2.4 mg weekly or placebo for 52 weeks. KCCQ-CSS improved by 16.6 points on semaglutide versus 8.7 on placebo, a 7.8-point placebo-adjusted gain. Six-minute walk distance gained 21.5 m versus placebo. Weight fell roughly 13% versus 2.6%. The companion STEP-HFpEF DM trial in patients with type 2 diabetes (Kosiborod et al. NEJM 2024, PMID 38587233) confirmed similar KCCQ and weight effects in the diabetic HFpEF population.
The two trials answer different questions. STEP-HFpEF was powered for symptom and weight endpoints, not for hard cardiovascular events. SUMMIT was powered for both. The KCCQ effects are similar in magnitude, the weight effects are similar in magnitude, but only SUMMIT has a positive primary cardiovascular event endpoint. That difference matters when a clinician is deciding between a GLP-1 monotherapy and a GIP/GLP-1 co-agonist for an obese HFpEF patient with active cardiovascular risk.
| Trial | Drug | N | Population | Duration | KCCQ-CSS effect | 6MWD effect | Weight | Hard CV endpoint |
|---|---|---|---|---|---|---|---|---|
| STEP-HFpEF | Semaglutide 2.4 mg | 529 | HFpEF + BMI 30+, no DM | 52 weeks | +7.8 vs PBO | +21.5 m | -13.3% | not powered |
| STEP-HFpEF DM | Semaglutide 2.4 mg | 616 | HFpEF + BMI 30+, T2D | 52 weeks | +7.3 vs PBO | similar | -9.8% | not powered |
| SUMMIT | Tirzepatide 15 mg | 731 | HFpEF + BMI 30+ | 52 weeks (KCCQ) / 104 weeks (CV) | +6.9 vs PBO | +18.3 m | -11.6 pp | HR 0.62 |
What this comparison does not establish: a head-to-head between the two drugs in HFpEF. There is no semaglutide-versus-tirzepatide trial in heart failure. The relative magnitudes look comparable on the symptom side; SUMMIT pulled ahead on the hard-outcome side primarily by being the trial designed to ask that question.
For the broader weight-loss head-to-head outside of heart failure, the tirzepatide vs semaglutide 2026 comparison covers the SURMOUNT-5 results and the body-weight delta. The pattern in heart failure mirrors the pattern in obesity: similar direction, edge to tirzepatide on absolute magnitude.
CKD subgroup and the kidney signal
About half of HFpEF patients have chronic kidney disease, and many of the drug classes that work in HFpEF require renal dose adjustment or worsen kidney function transiently. SUMMIT enrolled a population in which roughly 48% had baseline CKD (eGFR less than 60 mL/min/1.73 m^2 or evidence of albuminuria).
The CKD subgroup analysis (Butler et al. JACC 2025, PMID 40162940) showed that the cardiovascular event benefit and the KCCQ-CSS benefit held in patients with CKD. The relative risk reduction was numerically similar; the absolute risk reduction was larger in the CKD subgroup because the baseline event rate was higher. Tirzepatide also produced a placebo-adjusted increase in eGFR at 52 weeks when assessed by cystatin C, with a transient eGFR-creatinine dip at 12 weeks that resolved by 52 weeks in patients with CKD at baseline.
The eGFR signal is not the same as a clinical kidney outcome, and SUMMIT was not powered for renal endpoints. But the data are inconsistent with the worry that tirzepatide harms kidney function in obese HFpEF patients with baseline CKD. The signal points the opposite direction.
Tip: The eGFR-creatinine dip at 12 weeks is a known artifact of muscle-mass change with rapid weight loss. Researchers reading this evidence should weigh cystatin-C-based eGFR more heavily during the first six months of any GLP-1 or GIP/GLP-1 protocol in obese patients.
What SUMMIT does not establish
A clean read of the trial requires knowing where it stops.
- Lean HFpEF. SUMMIT enrolled only patients with BMI at or above 30 kg/m^2. The result does not extend to non-obese HFpEF, and the proposed mechanism (visceral and ectopic adipose unloading, LV mass remodeling, anti-inflammatory effect) is most plausible in the obese phenotype.
- HFrEF. The trial enrolled ejection fraction at or above 50%. Tirzepatide has not been tested in a Phase 3 trial for heart failure with reduced ejection fraction. The STEP-HFpEF and SUMMIT class signal does not transfer automatically; the underlying disease physiology differs.
- Versus SGLT2. SUMMIT used standard background therapy, which in 2024-2025 increasingly included SGLT2 inhibitors. The trial does not establish whether tirzepatide adds to SGLT2 in HFpEF or replaces it. Direct comparisons would require a different protocol.
- Off-target safety. Discontinuation due to gastrointestinal side effects was higher on tirzepatide than placebo, in line with prior SURMOUNT data. The SUMMIT cohort did not flag a new safety signal versus the obesity trials, but the trial was not designed to detect rare events.
- Lean-mass loss. Weight loss in SUMMIT included a fat-mass and lean-mass component, as in every GLP-1 obesity trial. The lean-mass implications for an older HFpEF cohort with baseline frailty are an open question the trial did not answer. For the broader picture on lean-mass preservation, see the GLP-1 muscle loss research read.
Where SUMMIT fits in the GIP/GLP-1 evidence base
Lay the SUMMIT result alongside the rest of the tirzepatide Phase 3 program and the shape of the molecule's clinical case becomes clearer.
| Indication | Trial | Status | Effect |
|---|---|---|---|
| Type 2 diabetes | SURPASS family | FDA approved (Mounjaro 2022) | A1c and weight superior to GLP-1 monotherapy |
| Obesity, no DM | SURMOUNT-1 (PMID 35658024) | FDA approved (Zepbound 2023) | -22.5% body weight at 72 weeks |
| Obesity vs semaglutide | SURMOUNT-5 | Read out 2024 | Tirzepatide superior on weight |
| Obstructive sleep apnea + obesity | SURMOUNT-OSA | FDA approved (Zepbound, December 2024) | AHI reduced up to 62.8% vs placebo |
| HFpEF + obesity | SUMMIT | Phase 3 positive 2024 | CV events HR 0.62; KCCQ +6.9 |
Read across that table: the tirzepatide Phase 3 program has now demonstrated benefit on three distinct cardiometabolic outcomes (weight, sleep apnea, HFpEF), with a fourth in long-term follow-up (cardiovascular outcomes in T2D via SURPASS-CVOT, expected through 2025-2026). For researchers tracking the GIP/GLP-1 class as a category, SUMMIT is the cleanest signal yet that incretin co-agonism produces structural and functional cardiovascular changes that outrun the weight-loss explanation.
The pattern is recognizable. The same molecule that drove the SURMOUNT-OSA AHI reduction is the same molecule that dropped LV mass and paracardiac adipose in SUMMIT. The hypothesis that a single mechanism (visceral and ectopic adipose unloading plus systemic anti-inflammatory effect) drives several of these wins is testable, and the substudies are increasingly testing it.
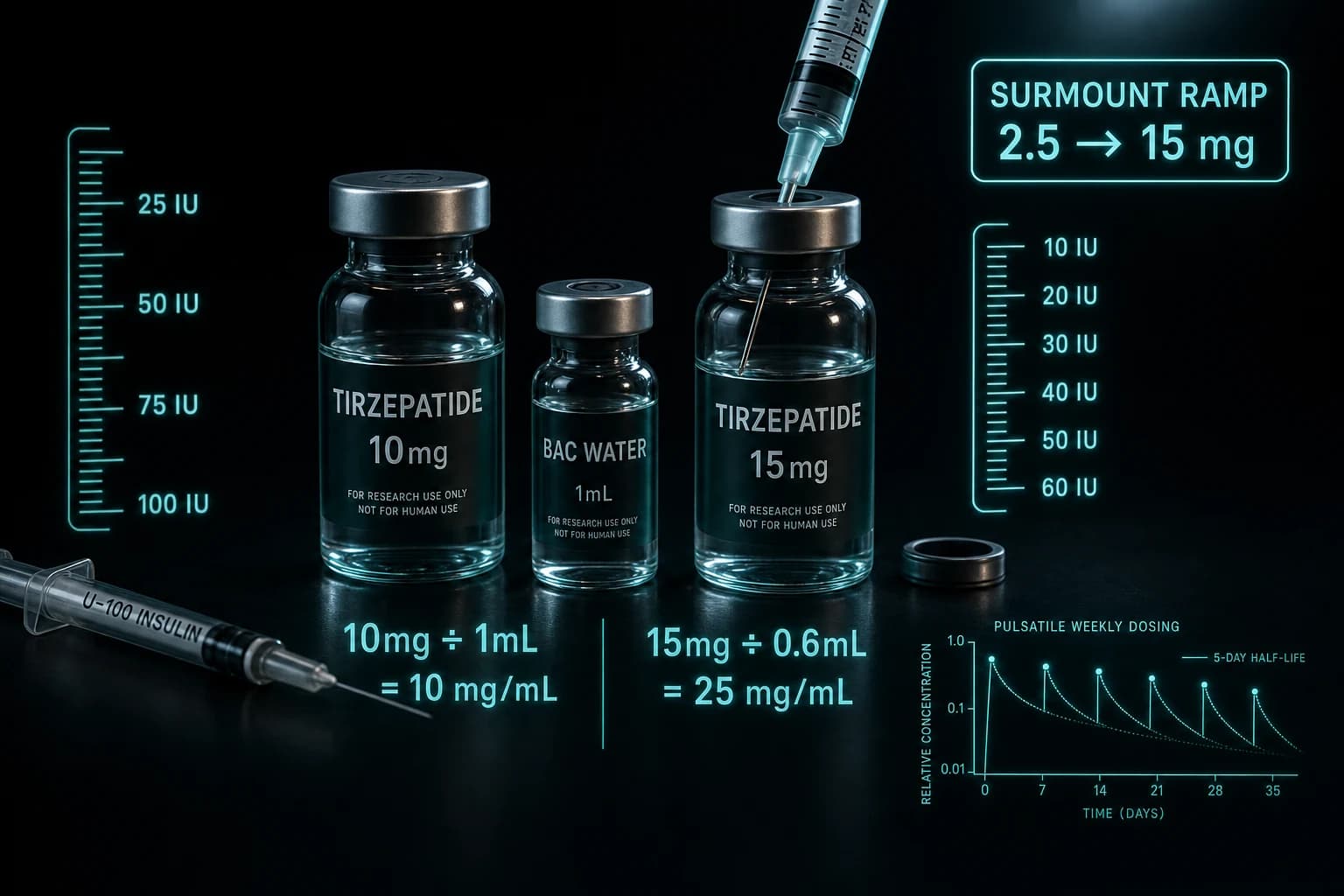
For the dosing math researchers actually use to run protocols at this dose range, the tirzepatide reconstitution chart for 10mg and 15mg vials covers the BAC water and IU calculations.
What this means for the research-use buyer
Tirzepatide is the same molecule whether sold under the FDA brand names (Mounjaro, Zepbound) or under research-use disclosures. The published Phase 3 data are the same. What differs is supply chain, formulation, and the regulatory frame around how a buyer can use it.
For research-use injectable supply with public per-batch certificates of analysis, Ascension Peptides is the partner we recommend, and code ENHANCED earns 50% off. For oral peptide protocols (not applicable to tirzepatide itself, which is injectable, but relevant to companion compounds), Limitless Biotech is the partner with code ENHANCED.
Two practical caveats worth surfacing before any reader operationalizes this. First, the trial used 15 mg as the maximum tolerated dose with the standard SURMOUNT titration. Researchers running a protocol that mirrors SUMMIT need the same titration ramp; the tirzepatide reconstitution math article covers the IU readouts. Second, SUMMIT enrolled patients on background heart-failure therapy. The cardiovascular benefit is on top of standard care, not as a replacement for it. None of this is a substitute for clinical management.
For researchers planning longer protocols, the stopping GLP-1s and weight regain research read covers what happens when an incretin protocol is discontinued, which has direct implications for how a SUMMIT-style protocol would need to be sustained.
FAQ
Is tirzepatide approved for HFpEF
As of 2026, no. SUMMIT is a positive Phase 3 trial, but FDA action on an HFpEF indication has not yet completed. Zepbound is approved for chronic weight management and for obstructive sleep apnea in adults with obesity (December 2024). Off-label prescribing for HFpEF in obese patients is happening clinically, but the formal label addition is pending.
Did SUMMIT replace SGLT2 inhibitors
No. SUMMIT used standard background therapy, which in many sites already included SGLT2 inhibitors. The trial does not establish whether tirzepatide is additive to SGLT2 inhibition or whether it is a substitute. The clean inference from SUMMIT is that tirzepatide adds clinical benefit on top of whatever was being prescribed at enrollment.
How big is a 6.9-point KCCQ-CSS gain
Clinically meaningful. The conventional threshold for a small but clinically important change in KCCQ-CSS is 5 points; 10 points is a moderate change; 20 is a large change. SUMMIT's 6.9-point placebo-adjusted gain at 52 weeks sits between the small and moderate thresholds and exceeds what most HFpEF drug trials have produced. STEP-HFpEF reported a similar 7.8-point gain, also in the small-to-moderate range.
Who should not be assumed to benefit
Lean HFpEF patients (BMI under 30) were not enrolled. Patients with HFrEF (ejection fraction under 50%) were not enrolled. Patients without an obesity-driven phenotype were not enrolled. The proposed mechanism (visceral adipose unloading, LV mass remodeling) is most plausible in the obese cohort the trial actually studied. Extending the result to other phenotypes is speculation, not evidence.
Does the cardiovascular event benefit require the full 15 mg dose
The trial titrated to maximum tolerated dose, which was 15 mg in most patients but lower in those who could not tolerate the highest titration step. The trial was not designed to compare doses; the Phase 3 effect should be read as the effect of the maximum-tolerated-dose titration ramp, not as a 15 mg-only effect.
Further reading
- Tirzepatide compound page
- Semaglutide compound page
- Tirzepatide vs semaglutide 2026 head-to-head
- Tirzepatide for sleep apnea: SURMOUNT-OSA read
- Tirzepatide reconstitution chart for 10mg and 15mg vials
- GLP-1 muscle loss research read
- Stopping GLP-1s: weight regain research
- Retatrutide explained: triple agonist Phase 2
Affiliate disclosure
We are an affiliate partner of Ascension Peptides and Limitless Biotech. Code ENHANCED earns us a commission on checkouts that originate from this site. The discount applies the same way regardless of how the buyer arrives at checkout. Our recommendations are based on what is publicly verifiable in COA libraries and in the Phase 3 published evidence; we would hold the same positions without the partnerships, but you should weigh the affiliate context as you would for any vendor review.
This article is for educational and research purposes only. Tirzepatide is FDA approved as Mounjaro for type 2 diabetes and as Zepbound for chronic weight management and for obstructive sleep apnea in adults with obesity. It is not approved for heart failure. None of the content above constitutes medical advice. Patients with heart failure should be managed by qualified clinicians; nothing in this read replaces that.