At a glance
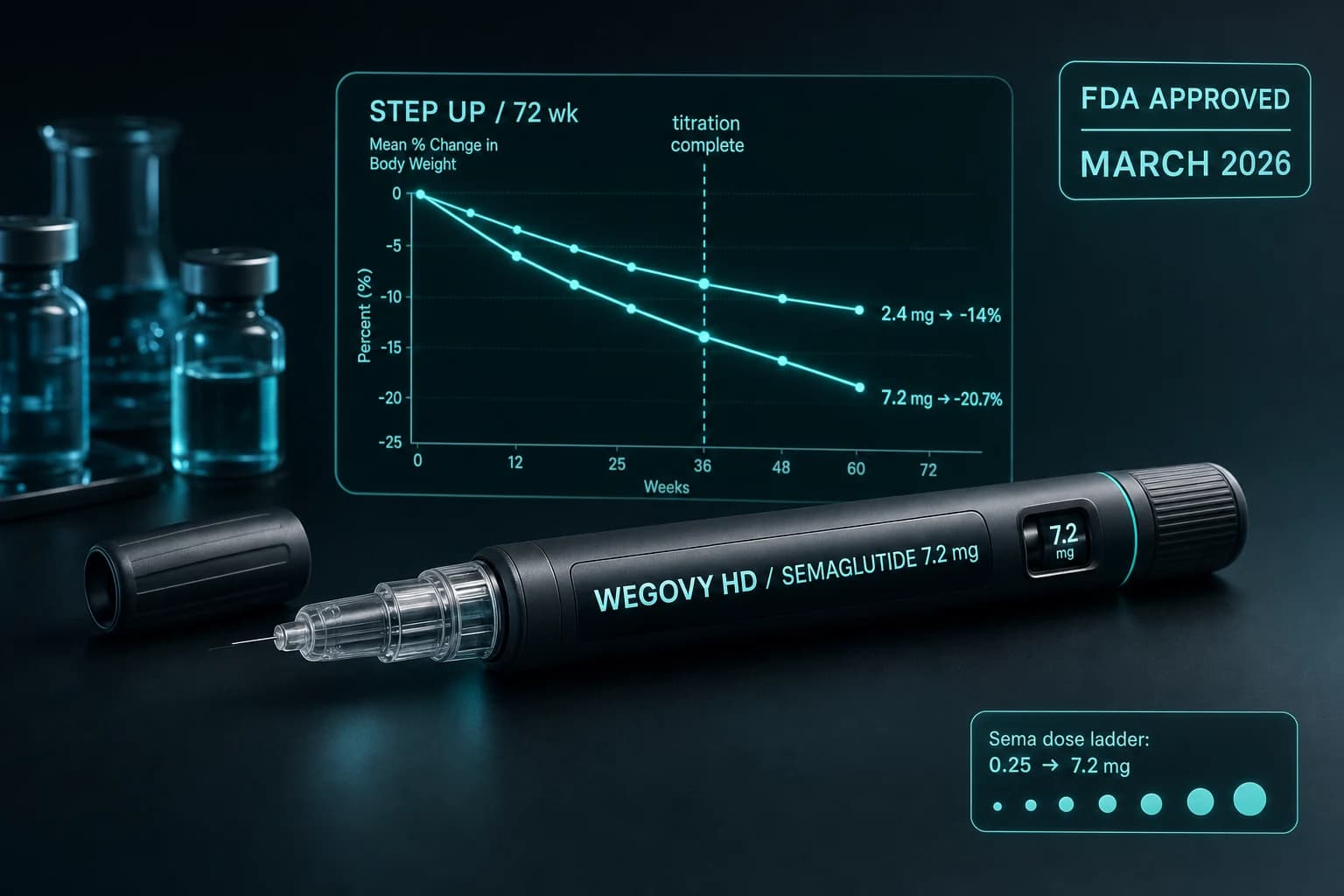
- Wegovy HD 7.2 mg was FDA approved on March 19, 2026 through the Commissioner's National Priority Voucher pilot program
- STEP UP Phase 3 (~1,400 adults, 72 wk): mean weight loss 20.7%, with 31.2% of participants reaching at least 25% weight loss
- Type 2 diabetes arm of STEP UP: 14.1% weight loss at 7.2 mg, the highest semaglutide result in the diabetes population
- Dose ladder adds two steps above standard Wegovy: 4.8 mg held 4 weeks, then 7.2 mg as maintenance, for a 25-week total titration
- Wegovy HD is for adults with obesity who tolerated 2.4 mg first and want or need more weight loss; jumping past the ladder is not how the trial was run
Wegovy HD (semaglutide 7.2 mg) is the first semaglutide formulation approved above the 2.4 mg dose. The FDA cleared it on March 19, 2026 through the Commissioner's National Priority Voucher pilot program, and the supporting Phase 3 trial, STEP UP, produced the highest weight loss result for any injectable semaglutide formulation to date: 20.7% mean reduction at 72 weeks in adults with obesity.
This article covers the STEP UP trial design and results, the new dose ladder, the side effect profile at 7.2 mg versus 2.4 mg, who the higher dose is appropriate for, and how it changes the GLP-1 dose conversation now that semaglutide and tirzepatide are within roughly two percentage points of each other at maximum dose.
STEP UP trial summary
The 72-week STEP UP Phase 3 trial evaluated weekly injectable semaglutide 7.2 mg compared to semaglutide 2.4 mg and placebo as adjuncts to lifestyle intervention in approximately 1,400 adults with obesity. The primary endpoint was mean change in body weight at week 72.
| Arm | Mean weight loss (72 wk) | At least 25% loss | Notable side effects |
|---|---|---|---|
| Placebo | ~-3% | low single digits | minimal |
| Semaglutide 2.4 mg | ~-14% | ~16% | nausea, diarrhea, constipation |
| Semaglutide 7.2 mg | -20.7% | 31.2% | same class, higher GI burden |
The 20.7% result is the highest published mean weight loss for any semaglutide product. The 31.2% rate of patients losing at least 25% of body weight is the high-bar response rate the trial program was designed to capture.
Bottom line: Tripling the semaglutide dose from 2.4 mg to 7.2 mg produces about 6 additional percentage points of weight loss over 72 weeks. The dose-response curve is still ascending at 2.4 mg, which is why STEP UP worked. The 7.2 mg ceiling does not yet match tirzepatide 15 mg, but the gap is now narrow.
A second STEP UP arm evaluated 7.2 mg in adults with obesity and type 2 diabetes. Mean weight loss in that arm was 14.1%, also the highest semaglutide weight-loss result in the diabetes population. For context, the prior best in T2D was the SUSTAIN program at 2.4 mg, which produced approximately 10% weight loss.
The new dose ladder
Standard Wegovy uses a 5-step titration over 17 weeks: 0.25, 0.5, 1.0, 1.7, 2.4 mg, each held for 4 weeks. Wegovy HD extends the ladder with two additional steps:
| Week range | Dose | Format |
|---|---|---|
| Weeks 1-4 | 0.25 mg | Standard Wegovy pen |
| Weeks 5-8 | 0.5 mg | Standard Wegovy pen |
| Weeks 9-12 | 1.0 mg | Standard Wegovy pen |
| Weeks 13-16 | 1.7 mg | Standard Wegovy pen |
| Weeks 17-20 | 2.4 mg | Standard Wegovy pen |
| Weeks 21-24 | 4.8 mg | Wegovy HD intermediate pen |
| Week 25 onward | 7.2 mg | Wegovy HD maintenance pen |
The full ramp from injection 1 to 7.2 mg maintenance is approximately 25 weeks. This is a long titration by intent. The slower escalation is the reason the side effect profile at 7.2 mg in STEP UP did not produce a dramatically higher discontinuation rate compared to 2.4 mg, despite tripling the active dose.
Patients who are already on standard Wegovy at 2.4 mg can transition through the 4.8 mg step rather than restarting at 0.25 mg. The 4.8 mg step is held for 4 weeks before escalating to 7.2 mg maintenance, so the practical transition for an existing 2.4 mg patient adds approximately 8 weeks before reaching the new maintenance.
The pen format matters operationally. Wegovy HD ships in a single-dose pen at the higher dose, distinct from the standard Wegovy multi-dose pen. The launch began in April 2026 in the United States.
Side effect profile at 7.2 mg
The side effect class for semaglutide remains the same at 7.2 mg as at 2.4 mg. The frequency and severity at each timepoint shift.
The most common dose-dependent events in STEP UP:
Nausea. Most frequent in the first 8 weeks of any new dose step. The 4.8 mg titration step in particular reportedly produced a transient bump in nausea reports before tolerance restored. By week 12 of maintenance at 7.2 mg, nausea rates approximated 2.4 mg patient rates from STEP 1.
Vomiting. Higher rate at 7.2 mg than 2.4 mg, particularly during the 4.8 → 7.2 mg escalation window. Most events were mild-to-moderate and did not require discontinuation.
Diarrhea and constipation. Both occurred at higher rates than 2.4 mg, with constipation generally more persistent than diarrhea over the trial duration.
Decreased appetite and reduced food intake. This is the mechanism, not a side effect, but it scales with dose. At 7.2 mg, patients in STEP UP reported substantially lower caloric intake than at 2.4 mg, which is part of how the additional weight loss is produced.
The trial's overall discontinuation rate due to adverse events was higher in the 7.2 mg arm than the 2.4 mg arm but not dramatically so. The Phase 3 program demonstrated that the longer titration ladder is the necessary structural element for tolerability.
For our deeper coverage of semaglutide discontinuation and what happens after stopping, see the stopping GLP-1 and weight regain research.
Where Wegovy HD sits versus the rest of the class
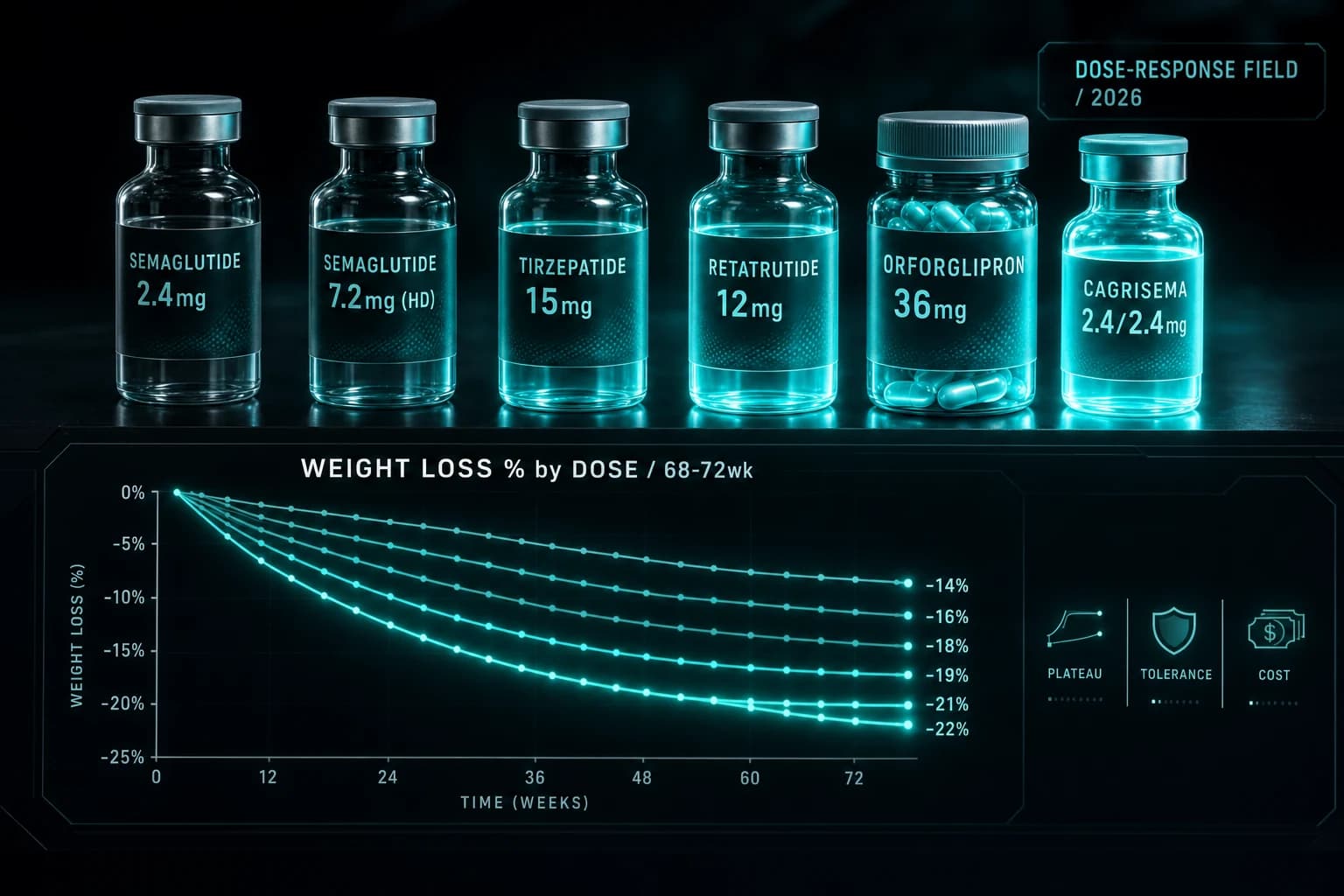
The arrival of 7.2 mg semaglutide closes most of the gap between semaglutide and tirzepatide at their top doses.
| Compound | Top approved dose | Trial | Top-dose weight loss | Duration |
|---|---|---|---|---|
| Semaglutide 7.2 mg | Wegovy HD | STEP UP | -20.7% | 72 wk |
| Tirzepatide 15 mg | Zepbound | SURMOUNT-1 | -22.5% | 72 wk |
| Retatrutide 12 mg | (not approved) | Phase 2 | -24.2% | 48 wk |
| CagriSema 2.4/2.4 mg | (filed) | REDEFINE-1 | -22.7% | 68 wk |
Tirzepatide still has the top number among approved monotherapies (22.5% vs 20.7%). The two-percentage-point gap is small, and the choice between semaglutide HD and tirzepatide 15 mg is now driven more by individual tolerance, prior treatment history, access, and cost than by the on-paper effect size.
CagriSema's 22.7% at 2.4/2.4 mg comes from a combination of cagrilintide and semaglutide, not from a higher semaglutide monotherapy dose. Retatrutide's 24.2% is the Phase 2 ceiling and not approved. The retatrutide vs tirzepatide vs semaglutide head-to-head and the GLP-1 dosing comparison 2026 cover the broader landscape.
For the prior best-in-class comparison, see tirzepatide vs semaglutide 2026.
Who should consider Wegovy HD
The HD label is for adults with obesity who are appropriate candidates for chronic weight management therapy. Within that label, the practical question is when a patient already on 2.4 mg should escalate to 7.2 mg.
A few cases where the escalation makes mechanistic sense:
Insufficient response at 2.4 mg. A patient who reaches 2.4 mg, holds it for several months, and has plateaued at a weight loss well below their goal is the cleanest case for escalation. The dose-response data shows continued response, not flat curve, between 2.4 mg and 7.2 mg.
Good tolerance at 2.4 mg. If 2.4 mg produced minimal GI side effects, the patient is likely to tolerate the 4.8 → 7.2 mg ramp. The patients who struggled at 2.4 mg generally are not the right candidates for 7.2 mg.
Type 2 diabetes patients seeking additional A1C reduction. The 7.2 mg dose in the diabetes arm produced larger A1C reductions than 2.4 mg in addition to greater weight loss. For patients whose diabetes is well-controlled on lower-dose GLP-1 but who want better weight outcomes, the HD dose is worth discussing with their clinician.
Cases where 7.2 mg is probably not the right move:
- Patients with significant GI intolerance at lower doses
- Patients close to their weight goal where the marginal benefit is small
- Patients with risk factors for pancreatitis or gallbladder disease, where higher GLP-1 exposure may warrant additional caution
- Patients in early titration who have not stabilized at 2.4 mg
The escalation decision is a clinician decision with the patient. Wegovy HD is not appropriate as a starter dose, and direct initiation at 7.2 mg is not how STEP UP was run or how the label is written.
Bottom line: Good candidates for Wegovy HD have stable tolerance at 2.4 mg, insufficient weight loss to reach their goal, and time to run a 25-week titration. Patients struggling with GI tolerance at 2.4 mg are typically not the right candidates for the HD step.
Cost and access
Wegovy HD is a separate product from standard Wegovy and is priced as a premium tier. Insurance coverage in the United States for the HD formulation rolled out gradually through Q2 2026 with formulary placement varying by payer. Patients escalating from 2.4 mg should expect a prior authorization conversation similar to the original Wegovy approval.
The list price of the HD pen is higher than the 2.4 mg pen, reflecting both the higher active dose and the new product positioning. Manufacturer coupons and patient assistance programs may apply for eligible patients, mirroring the access pathway for standard Wegovy.
For research-grade injectable semaglutide vials at any dose level, Ascension Peptides ships 5 mg and larger vials with public per-batch COAs and 50% off using code ENHANCED. The semaglutide reconstitution chart covers dose-by-volume math for research-grade vials at standard 5 mg sizes.
Compounded vs branded high-dose
The compounded peptide market has been selling research-grade semaglutide at doses functionally equivalent to or above 7.2 mg for several years. The Wegovy HD approval validates the dose-response concept above 2.4 mg with a Phase 3 trial.
A few practical distinctions remain:
-
Titration is the entire safety story. The STEP UP trial's tolerable side effect profile at 7.2 mg depends on the 25-week titration. A compounded 7.2 mg dose used without titration is a different risk profile from the branded HD protocol.
-
Branded HD has direct Phase 3 efficacy data. Compounded high-dose semaglutide does not have direct Phase 3 confirmation at its specific dose. Mechanism extrapolation is reasonable but not equivalent to a randomized trial.
-
The FDA 503A compounding picture is evolving. Semaglutide is currently on the FDA shortage-driven 503A compounding allowance, which means compounding pharmacies can produce it for individual patient prescriptions. The shortage status and the regulatory environment around obesity drug compounding continue to change. For broader context, see the FDA 503A peptide compounding review July 2026.
For research-grade applications where dose flexibility and per-batch documentation matter, see the best legit peptide vendors 2026 ranking and the retatrutide where-to-buy 2026 guide for analogous standards.
FAQ
Should I switch from Wegovy 2.4 mg to Wegovy HD 7.2 mg?
The answer depends on three factors: response at 2.4 mg, tolerance at 2.4 mg, and weight goal. Patients with good tolerance at 2.4 mg but insufficient weight loss to reach their goal are the clearest candidates. Patients close to goal or struggling with GI tolerance at 2.4 mg are typically not the right candidates. The decision should involve a clinician evaluation, not a self-escalation.
How long does it take to reach 7.2 mg from scratch?
Approximately 25 weeks. The dose ladder is 0.25, 0.5, 1.0, 1.7, 2.4, 4.8, 7.2 mg, with each step held for 4 weeks. The slower titration is intentional and is what makes the 7.2 mg dose tolerable in the trial population.
Is Wegovy HD better than tirzepatide 15 mg?
Tirzepatide 15 mg produced 22.5% weight loss in SURMOUNT-1 over 72 weeks. Wegovy HD 7.2 mg produced 20.7% in STEP UP over 72 weeks. Tirzepatide remains the higher number among approved monotherapies, but the gap is small enough that individual tolerance, access, and prior treatment history matter more than the on-paper difference. There is no head-to-head trial between Wegovy HD and tirzepatide 15 mg.
What side effects should I expect transitioning from 2.4 mg to 7.2 mg?
The most common transition-window events are nausea, vomiting, and decreased appetite, particularly during the 4.8 → 7.2 mg escalation step. Most events are mild-to-moderate and resolve within 4-6 weeks of the new dose. Patients who had GI symptoms at 2.4 mg tend to have them again at the new escalation step. Patients who tolerated 2.4 mg easily tend to tolerate the HD steps similarly.
Can I jump straight from 0.25 mg or 1.0 mg to 7.2 mg?
No. The full dose ladder is required for tolerability and was the structure of the STEP UP Phase 3 trial. Direct initiation at 7.2 mg is not how the trial was run, not how the label is written, and would produce significantly worse GI tolerance.
Is Wegovy HD approved for type 2 diabetes?
Wegovy HD is approved for chronic weight management in adults with obesity. The STEP UP trial included a type 2 diabetes arm with 14.1% weight loss results, but the indication is for weight management, not diabetes. Ozempic (semaglutide for T2D) is dosed up to 2 mg weekly and is a separate product. As of May 2026, the 7.2 mg dose is not approved for diabetes-specific indications.
What is the FDA Commissioner's National Priority Voucher program?
The Commissioner's National Priority Voucher is a 2026 FDA accelerated review pathway designed to compress regulatory review for products addressing priority public health needs. Wegovy HD was the first GLP-1 approved through this pathway. Approval timelines under the voucher are meaningfully shorter than standard review.
Will Wegovy HD reach my pharmacy soon?
The US launch began in April 2026 in a single-dose pen format. Pharmacy availability has been rolling through Q2 2026 with payer formulary placement varying. Patients should check with their pharmacy and insurer before assuming the HD product is available at their counter.
Further reading
- GLP-1 dosing comparison 2026: from 2.4 mg to 7.2 mg and beyond
- Tirzepatide vs Semaglutide 2026 head-to-head
- Retatrutide vs Tirzepatide vs Semaglutide
- Semaglutide reconstitution chart 5mg
- Stopping GLP-1 and weight regain research
- GLP-1 muscle loss research
- CagriSema REDEFINE Phase 3 guide
- FDA 503A peptide compounding review July 2026
- Best legit peptide vendors 2026
This article is for educational and research purposes only. Wegovy and Wegovy HD are FDA-approved formulations of semaglutide for chronic weight management. None of the content above constitutes medical advice. Dose escalation decisions should be made by qualified clinicians with patients. Consult a clinician for individual medical questions about weight management therapy.