At a glance
- Standard research protocol: 250-500 mcg daily subcutaneous, fasted morning, 12-week cycles
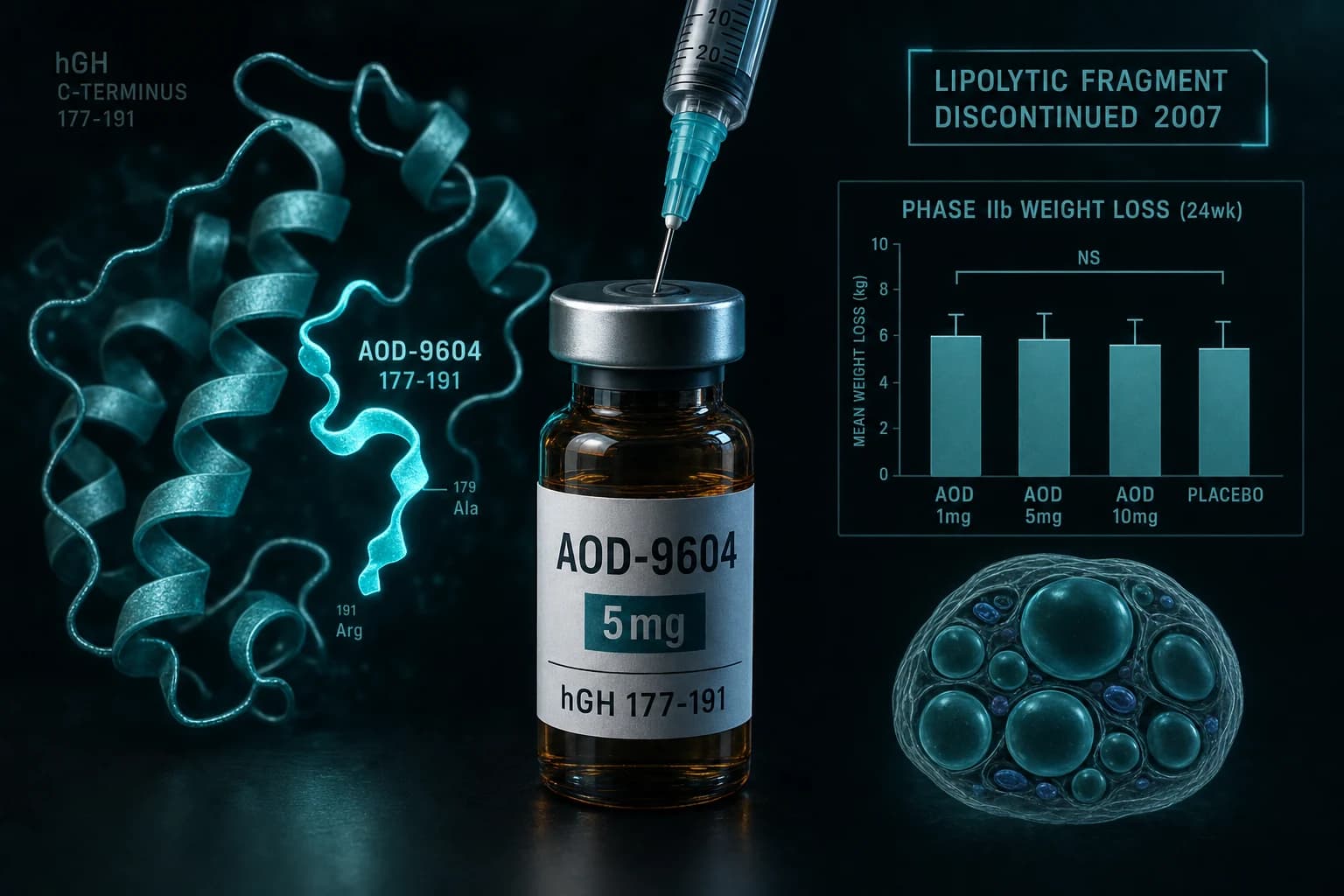
- AOD-9604 is the C-terminal hGH fragment (residues 177-191) that retains the lipolytic activity
- Phase IIb (536 subjects, 24 weeks) failed to show statistical significance versus placebo, leading to discontinuation in 2007
- Mechanism: stimulates lipolysis and fatty acid oxidation in adipocytes without altering IGF-1 or growth hormone axis
- Six human trials with 900+ subjects established safety; the efficacy signal in obese humans was inconsistent
If you are looking up an AOD-9604 dose, you are most likely either evaluating it as a fat-loss adjunct to a growth-hormone-secretagogue stack or trying to understand why a peptide with a clean mechanism failed in clinical trials and is now sold as a research compound.
This guide covers the standard research protocol used across animal-model adaptations and forum-documented protocols, the actual mechanism (it is the C-terminal lipolytic fragment of human growth hormone), and the Phase IIb data that ended the original commercial development program in 2007.
The standard research protocol
The convergent research-grade protocol is 250-500 mcg daily, subcutaneous, fasted morning, for 12-week cycles.
| Phase | Dose | Frequency | Timing | Duration |
|---|---|---|---|---|
| Standard | 300 mcg | Once daily, SC | Morning, fasted | 12 weeks |
| Higher-end | 500 mcg | Once daily, SC | Morning, fasted | 12 weeks |
| Twice-daily | 150 mcg AM + 150 mcg PM | Twice daily | Fasted both | Optional |
The fasted-morning timing comes from the mechanism. AOD-9604 is positioned as a lipolytic compound that drives fatty acid mobilization from adipose tissue. The effect is amplified when injected in a fasted state, before insulin and the post-meal anabolic environment dampens lipolysis. Researchers running it for fat-loss-adjacent endpoints typically inject 30 minutes before fasted cardio or before breakfast on rest days.
The 12-week cycle bound is mostly inherited from the original Metabolic Pharmaceuticals trial designs and the convention that emerged in research forums. There is no specific safety reason to cap at 12 weeks; it is just the duration most-tested in the published literature.
What AOD-9604 actually is
AOD-9604 is a 16-amino-acid synthetic peptide corresponding to residues 177-191 of human growth hormone, with a tyrosine added to the N-terminus for stability. The fragment retains the lipolytic activity of full hGH but lacks the receptor binding regions responsible for the growth-promoting and IGF-1-stimulating effects.
In other words: it is the part of growth hormone that drives fat metabolism, separated from the part that drives growth. The original development hypothesis was that this would deliver hGH-like fat-loss effects without the IGF-1 elevation, glucose disruption, or growth-promoting risk profile of full-length hGH therapy.
The mechanistic work supporting this came from Heffernan et al., Diabetes, 2001, which showed AOD-9604 increased fat oxidation and produced weight loss in obese mice through mechanisms partly independent of the growth hormone receptor. Earlier work by Ng et al., Horm Res, 2000 characterized the metabolic effects of AOD9604 specifically as a synthetic lipolytic domain.
For mechanistic depth on adipose lipolysis and how AOD-9604 fits the broader landscape of growth-hormone-related fat-loss compounds, the tesamorelin compound guide covers a related but distinct mechanism (GHRH analog that drives endogenous GH release rather than acting as a fragment), and the CJC-1295 + Ipamorelin protocol covers the secretagogue alternative.
What the human trial data actually shows
This is where AOD-9604 stops looking great on paper. The mouse data was strong. The human data was inconsistent.
AOD-9604 completed six human clinical trials with over 900 participants. The trial program was sponsored by Metabolic Pharmaceuticals (later Calzada Limited) and ran through the early-to-mid 2000s.
| Trial | Subjects | Duration | Dose | Outcome |
|---|---|---|---|---|
| Pilot IV/oral | small (<100) | short | various | Safety, PK established |
| Phase IIa oral | ~300 | 12 wk | 1 mg | Some weight-loss signal |
| Phase IIb oral | 536 | 24 wk | up to 30 mg | Did not reach statistical significance vs placebo |
The 12-week trial reportedly showed average weight loss of 2.6 kg in the AOD arm versus 0.8 kg in placebo, a statistically meaningful but small effect. The dose-response was not clean: the highest dose (10 mg in the early arm) did not produce more weight loss than the 1 mg dose.
The 24-week Phase IIb trial with 536 subjects was designed as the pivotal efficacy study. It did not reach statistical significance versus placebo. Metabolic Pharmaceuticals discontinued AOD-9604 development in 2007 on the basis of that result.
For a full review of AOD-9604's place in the pre-GLP-1-class obesity drug development landscape, Hofbauer et al., World J Diabetes, 2014 covers the central and peripheral molecular targets for anti-obesity pharmacotherapy with AOD-9604 included as a discontinued candidate. Hainer and Aldhoon-Hainerová, Obesity Pharmacotherapy review, 2013 discusses the same compound in the broader obesity pharmacotherapy context.
Why the disconnect between mouse and human?
A few possibilities, none confirmed:
-
Species differences in lipolytic receptors. The β3-adrenergic receptor profile that drives lipolysis in rodents differs from humans. Compounds that produce strong lipolytic effects in obese mice often produce smaller effects in humans. This is the same pattern that ended development for several rodent-validated adipose-targeting compounds.
-
Dose-finding may not have caught the right window. The Phase IIb trial used oral dosing up to 30 mg. Oral bioavailability of peptides is generally poor. The injectable doses where mechanism studies were positive were not directly tested in the pivotal trial.
-
Placebo effect in obesity trials is high. A 2-3 kg placebo arm weight loss over 24 weeks is normal for diet-controlled obesity trials. Beating placebo by a meaningful margin requires a strong drug effect, and AOD-9604 did not deliver that margin.
The compound was never reformulated as an injectable for human pivotal testing after the oral 24-week failure. The development decision was to move on rather than rerun with a different route, which closed the chapter on AOD-9604 as a commercial obesity drug.
Reconstitution math
Most research-grade AOD-9604 ships as 5 mg lyophilized vials.
| Vial mass | Bacteriostatic water added | Concentration | 0.1 mL draw |
|---|---|---|---|
| 5 mg | 1.0 mL | 5,000 mcg/mL | 500 mcg |
| 5 mg | 2.0 mL | 2,500 mcg/mL | 250 mcg |
| 5 mg | 5.0 mL | 1,000 mcg/mL | 100 mcg |
For a 300-500 mcg daily dose, 5 mg into 1 mL bacteriostatic water is the cleanest. A draw of 0.06-0.10 mL on a U-100 insulin syringe (6-10 IU) delivers 300-500 mcg. The vial yields 10-16 doses depending on the target dose.
If you want finer control or smaller doses (250 mcg), reconstitute with 2 mL. The same 0.10 mL draw now delivers 250 mcg, and the vial yields 20 doses.
The reconstitution calculator handles arbitrary vial sizes and target doses if your vial is not 5 mg.
Safety and what the trials reported
In all six published human trials, AOD-9604 was reported as safe and well tolerated at the doses tested. The compound did not raise IGF-1 levels (this was the headline mechanistic claim), did not produce dose-dependent glucose disruption, and did not produce growth-promoting effects on bones or organs.
Reported side effects in the human trials were minimal and not different from placebo at the doses tested. This is part of why the safety case for AOD-9604 has held up even though the efficacy case did not.
What the safety database does not cover: long-term injectable use in humans at higher doses than the oral pivotal trial. Researchers running daily injectable AOD-9604 are extrapolating from the oral safety database, which is reasonable but not direct evidence.
Where AOD-9604 fits in 2026
If the goal is fat loss with a clean mechanism and a strong human trial database, AOD-9604 is not the right tool. The tirzepatide vs semaglutide head-to-head, the retatrutide reconstitution guide, and the orforglipron Phase 3 evidence cover the GLP-1-class compounds with proven clinical efficacy.
If the goal is mechanism diversity in a fat-loss research stack (running AOD-9604 alongside growth-hormone secretagogues like CJC-1295/Ipamorelin to layer different lipolytic mechanisms), AOD-9604 is a defensible research lever with a clean safety profile and a published mechanism. The trade-off is that the human efficacy signal in obese subjects was modest in the trials that did show effects and absent in the pivotal Phase IIb.
The honest summary: AOD-9604 is a research-grade compound with strong mouse data, mixed human data, and a Phase IIb that did not work. It is not a substitute for the GLP-1 class. It is a niche tool for researchers running mechanism-comparative work or fat-loss-adjacent stacks.
Sourcing
Research-grade AOD-9604 is sold as injectable lyophilized vials. Ascension Peptides carries it in the injectable catalog with 50% off using code ENHANCED. For our broader sourcing analysis, the best legit peptide vendors 2026 ranking covers vendor-by-vendor COA standards.
For the closest mechanistic alternatives that did reach approval, the tesamorelin compound guide covers the GHRH analog that targets visceral adipose tissue and is FDA-approved for HIV-associated lipodystrophy.
FAQ
What is the standard AOD-9604 research dose?
The convergent research-grade protocol is 250-500 mcg daily, subcutaneous, fasted morning, for 12-week cycles. The 300-500 mcg range is the most common starting point. There is no clean dose-response curve from the human trial data, so this protocol is partly inherited from the early oral trials and partly from animal-model adaptation.
Did AOD-9604 actually work in humans?
The early Phase IIa trial showed modest weight loss (2.6 kg vs 0.8 kg placebo at 12 weeks). The pivotal Phase IIb trial with 536 subjects over 24 weeks did not reach statistical significance versus placebo. Metabolic Pharmaceuticals discontinued development in 2007. The compound is research-grade only and is not approved by any major health authority for any indication.
Why does AOD-9604 not raise IGF-1?
AOD-9604 is the C-terminal fragment of human growth hormone (residues 177-191) and lacks the N-terminal regions that bind the growth hormone receptor in liver to drive IGF-1 production. It retains the lipolytic activity that maps to a different mechanism in adipose tissue. This was the original design hypothesis and it has held up across the published mechanism work (Heffernan et al., 2001).
Should I run AOD-9604 fasted?
Yes, fasted morning dosing is the convention. The lipolytic mechanism is amplified when insulin levels are low, which is why most research protocols inject 30 minutes before fasted cardio or before the first meal of the day. Post-meal injection puts AOD-9604 into a high-insulin environment that is less favorable for lipolysis.
Can I stack AOD-9604 with CJC-1295 / Ipamorelin?
Yes, this is a common research stack. AOD-9604 targets adipose lipolysis directly. CJC-1295 + Ipamorelin drives endogenous GH release, which has its own lipolytic effects through different mechanisms. Stacking the two addresses fat loss through complementary pathways. The CJC-1295 + Ipamorelin protocol covers the secretagogue side of this stack in detail.
Is AOD-9604 FDA approved for anything?
No. AOD-9604 is not approved for any indication. It was developed as an obesity compound through Phase IIb and discontinued in 2007 after the pivotal trial failed. Despite occasional off-label use claims, no major regulatory authority has approved AOD-9604.
Further reading
- Tesamorelin: visceral fat GHRH peptide guide
- CJC-1295 + Ipamorelin stack protocol
- Tirzepatide vs Semaglutide head-to-head 2026
- Best legit peptide vendors 2026
- Reconstitution Calculator
- AOD-9604 compound guide
This article is for educational and research purposes only. AOD-9604 is sold under research-use disclosures and is not approved by the FDA or any other major health authority for any indication. The Phase IIb pivotal obesity trial did not meet its primary endpoint, and the compound was discontinued in 2007. None of the dosing protocols described should be interpreted as medical advice. Consult a qualified clinician for individual medical questions.