At a glance
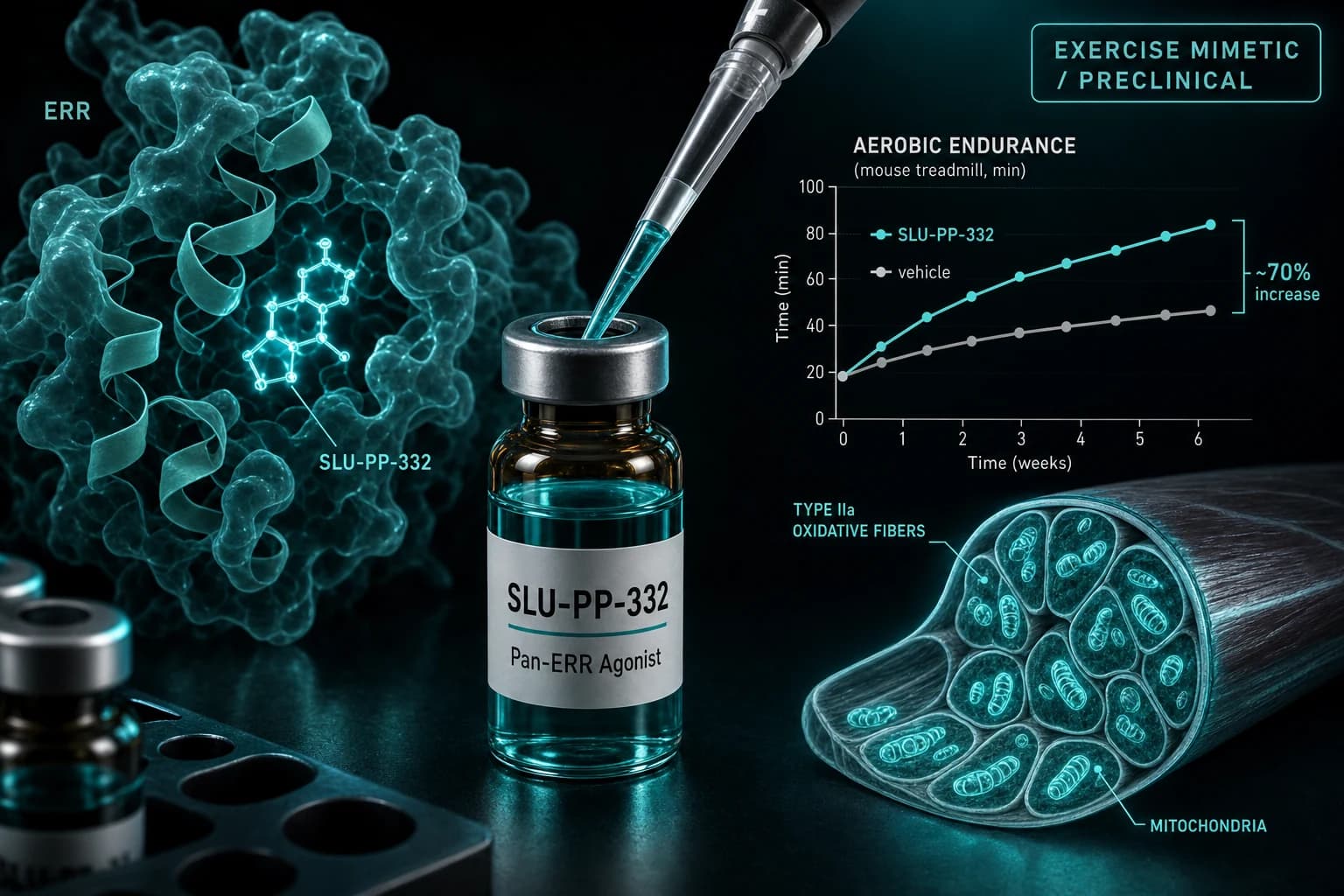
- Published preclinical dose: 50 mg/kg IP daily in mice (Billon et al., 2023)
- Oral bioavailability is poor; SLU-PP-915 is the orally bioavailable follow-up analog from the same lab
- Mechanism: pan-ERRα/β/γ agonist that increases mitochondrial biogenesis, fatty acid oxidation, and type IIa muscle fibers
- Mouse treadmill endurance improved ~70% versus vehicle in the original Nature Metabolism paper
- No human trials have been published as of May 2026; all dosing claims for human research are extrapolation
If you are looking up a SLU-PP-332 dose, you have probably seen the headlines about a "pill that mimics exercise" and want to translate that to a research protocol. The honest answer is that this is mostly preclinical, the published data is in mice, and the dose-translation question is harder than it looks because the original SLU-PP-332 has poor oral bioavailability.
This guide covers what the published literature actually says about SLU-PP-332 dosing, why the orally bioavailable follow-up analog (SLU-PP-915) exists, and what the practical implications are for researchers running this compound in 2026.
What the published data actually shows
The headline paper is Billon et al., Nature Metabolism, 2024 from Tom Burris's lab at the University of Florida (originally Saint Louis University, hence the "SLU" prefix). The published mouse study used:
| Parameter | Published protocol |
|---|---|
| Compound | SLU-PP-332 |
| Route | Intraperitoneal (IP) injection |
| Dose | 50 mg/kg |
| Frequency | Once daily |
| Duration | 4 weeks |
| Vehicle | DMSO + corn oil |
| Result | ~70% increase in treadmill running time vs vehicle |
The mouse data was striking. The compound increased oxidative type IIa muscle fibers, mitochondrial biogenesis, and fatty acid oxidation. Endurance capacity improved meaningfully. Body weight in obese mice trended down. The paper was rightly treated as a major preclinical finding.
What the paper did not establish: a human dose. There are no published human trials of SLU-PP-332 as of May 2026.
Why oral dosing is the practical problem
SLU-PP-332 has poor oral bioavailability. This is documented in the follow-up work from the same lab, which developed SLU-PP-915 (Billon et al., 2024) specifically as an orally bioavailable pan-ERR agonist analog. The headline of that follow-up paper is essentially: "we made one that works orally." The implication is that the parent SLU-PP-332 does not.
This creates a problem for research-grade vendors selling SLU-PP-332 as an oral capsule. The published data on the parent compound was generated by IP injection. The forum-reported "SLU-PP-332 oral capsule, 25-50 mg once daily" protocol is extrapolation from the mouse IP dose using body-surface-area scaling, applied to a route the original compound was not optimized for.
If you are evaluating an oral SLU-PP-332 product, two reasonable questions to ask:
- Is this actually SLU-PP-332 or is it the SLU-PP-915 analog being sold under the SLU-PP-332 name? The vendor's COA should answer this. The compounds are structurally distinct.
- What is the formulation strategy for oral absorption? SLU-PP-332 in a plain capsule is unlikely to reach systemic levels comparable to the published mouse IP dose. Lipid-based formulations or other absorption enhancers may be in use.
The cleanest answer for a researcher who wants to replicate the mouse data is to use injectable SLU-PP-332 if available. The cleanest answer for a researcher who wants oral absorption with the same mechanism is to look for SLU-PP-915 specifically.
Theoretical dose translation
If you are translating the mouse dose to a human research dose using standard body-surface-area conversion (Reagan-Shaw et al., FASEB J, 2008):
Mouse 50 mg/kg → Human equivalent ≈ 4 mg/kg
For a 70 kg researcher, this would translate to approximately 280 mg per day by IP injection. This is the theoretical translation, not a validated human research dose.
The forum-reported oral dosing of 25-50 mg per day for SLU-PP-332 capsules is an order of magnitude below this translation, which makes mechanistic sense only if oral absorption is poor (in which case the dose may not be reaching meaningful systemic levels) or if the compound is actually the more bioavailable SLU-PP-915 analog.
This is a research compound. The honest framing is that any dose in humans is extrapolation, no published trials exist, and the route mismatch between the published data (IP injection in mice) and the available retail format (oral capsule for humans) is unresolved.
Mechanism in one paragraph
SLU-PP-332 is a synthetic pan-agonist of the estrogen-related receptors (ERRα, ERRβ, ERRγ). The ERRs are nuclear receptors that regulate genes involved in mitochondrial biogenesis, fatty acid oxidation, and oxidative metabolism. Activating the ERRs in skeletal muscle drives a transcriptional program that resembles what endurance training produces: more mitochondria, more type IIa oxidative fibers, increased fatty acid oxidation capacity. This is the basis for the "exercise mimetic" framing.
For mechanistic depth, Xu et al., Cell, 2023 showed SLU-PP-332 alleviates metabolic syndrome features in obese mice, and Wei et al., Circulation, 2023 showed pan-ERR agonists improve cardiac function in heart failure mouse models. Tarry-Adkins et al., 2023 extended the mechanism to renal mitochondrial function in aging kidney models.
SLU-PP-332 vs other "exercise mimetic" compounds
| Compound | Mechanism | Stage of evidence | Practical use |
|---|---|---|---|
| SLU-PP-332 | Pan-ERR agonist | Preclinical (mouse) | Research only, IP route in published data |
| SLU-PP-915 | Pan-ERR agonist | Preclinical (mouse) | Research only, orally bioavailable analog |
| MOTS-c | Mitochondrial-derived peptide | Preclinical + early human | Research, injectable |
| 5-Amino-1MQ | NNMT inhibitor | Preclinical primarily | Research, oral |
| AICAR | AMPK activator | Mostly preclinical | Banned by WADA, not retail-available |
| GW501516 | PPARδ agonist | Preclinical, terminated for cancer findings | Banned, withdrawn from research |
| Tesofensine | Triple monoamine | Phase 2 obesity | Research, oral |
The "exercise mimetic" category has a checkered history. GW501516 (cardarine) reached late-stage development before being terminated for cancer findings in long-term rodent studies. AICAR is on the WADA prohibited list. SLU-PP-332 is the newer entrant with cleaner preclinical data, but the human safety database does not exist yet.
For our coverage of related mitochondrial mechanisms, the MOTS-c mitochondrial exercise peptide guide covers a different mitochondrial-targeting peptide, and the 5-Amino-1MQ NNMT inhibitor research review covers a different metabolic-modulating oral compound with similar research positioning.
Safety: what the preclinical data shows
In the mouse studies, SLU-PP-332 was reported as well tolerated at the 50 mg/kg IP dose. Body weight, food intake, and behavior remained normal. The published mouse work has not flagged red-flag findings.
What the safety database does not cover:
- Long-term exposure beyond the 4-week mouse studies
- Higher doses in humans
- Interaction with other ERR pathway modulators
- Any human safety data at all
For the researcher: this is an early-stage compound. Side effect profile in humans is unknown. The conservative position is that running this compound assumes the same risk profile as any other research compound with no human trial database.
What about WADA?
The original Billon paper explicitly framed SLU-PP-332 as a potential exercise mimetic, and the World Anti-Doping Agency has addressed exercise mimetics under the S2 prohibited substances category. Boldea et al., Drug Test Anal, 2025 published metabolite analysis specifically for doping-control purposes, indicating regulatory bodies are tracking the compound for athlete-testing applications.
Researchers in WADA-tested athlete pools should not use SLU-PP-332. Detection methods exist and are validated.
Sourcing
Research-grade SLU-PP-332 is sold by some peptide vendors as oral capsules. Given the route-of-administration concerns described above, COA verification matters more for SLU-PP-332 than for most retail research compounds. The best legit peptide vendors 2026 ranking covers what to look for in a vendor's purity documentation.
For oral metabolic compounds with clearer published evidence at the same retail format, Limitless Biotech carries 5-Amino-1MQ and tesofensine with code ENHANCED.
FAQ
What is the standard SLU-PP-332 research dose?
There is no standard human research dose. The published mouse data used 50 mg/kg IP injection daily. Forum-reported oral dosing for human research is 25-50 mg per day, which is an extrapolation rather than a validated protocol. Because the parent compound has poor oral bioavailability, the relationship between an oral retail dose and the systemic exposure that drives the published preclinical effects is unclear.
Why is SLU-PP-915 better than SLU-PP-332 if both are pan-ERR agonists?
SLU-PP-915 was specifically developed by the Burris lab as the orally bioavailable analog of SLU-PP-332. The original SLU-PP-332 has poor oral absorption, which limits its usefulness as a retail oral product. SLU-PP-915 retains the pan-ERR agonist mechanism with significantly improved oral bioavailability. If a retail product is targeting oral administration, SLU-PP-915 is the mechanistically appropriate choice.
Has SLU-PP-332 been tested in humans?
No published human trials exist as of May 2026. All available data is preclinical (mouse and cell-based). Human safety, efficacy, and pharmacokinetics are unknown. The compound is research-grade only.
Is SLU-PP-332 banned in sport?
The World Anti-Doping Agency tracks exercise mimetics under the S2 prohibited substances category. Doping-control metabolite analysis for SLU-PP-332 has been published (Boldea et al., 2025). Athletes in WADA-tested pools should not use SLU-PP-332.
Can I run SLU-PP-332 with MOTS-c or 5-Amino-1MQ?
Mechanistically, these compounds target different aspects of mitochondrial and metabolic regulation. SLU-PP-332 is a pan-ERR agonist, MOTS-c is a mitochondrial-derived peptide that acts through different pathways, and 5-Amino-1MQ is an NNMT inhibitor. There is no published evidence on combined dosing in either animals or humans. Researchers running stacks here are operating beyond what the literature has tested.
Further reading
- MOTS-c mitochondrial exercise peptide guide
- 5-Amino-1MQ NNMT inhibitor research review
- SS-31 (Elamipretide) FDA-approved mitochondrial peptide
- Best legit peptide vendors 2026
- Tesofensine dosage guide 2026
This article is for educational and research purposes only. SLU-PP-332 is a research compound with no published human trials and no FDA-approved indication. The dosing translation from the published mouse data to human research dosing is extrapolation, not validated protocol. Athletes in WADA-compliant testing pools should not use SLU-PP-332. Consult a qualified clinician for individual medical questions.