At a glance
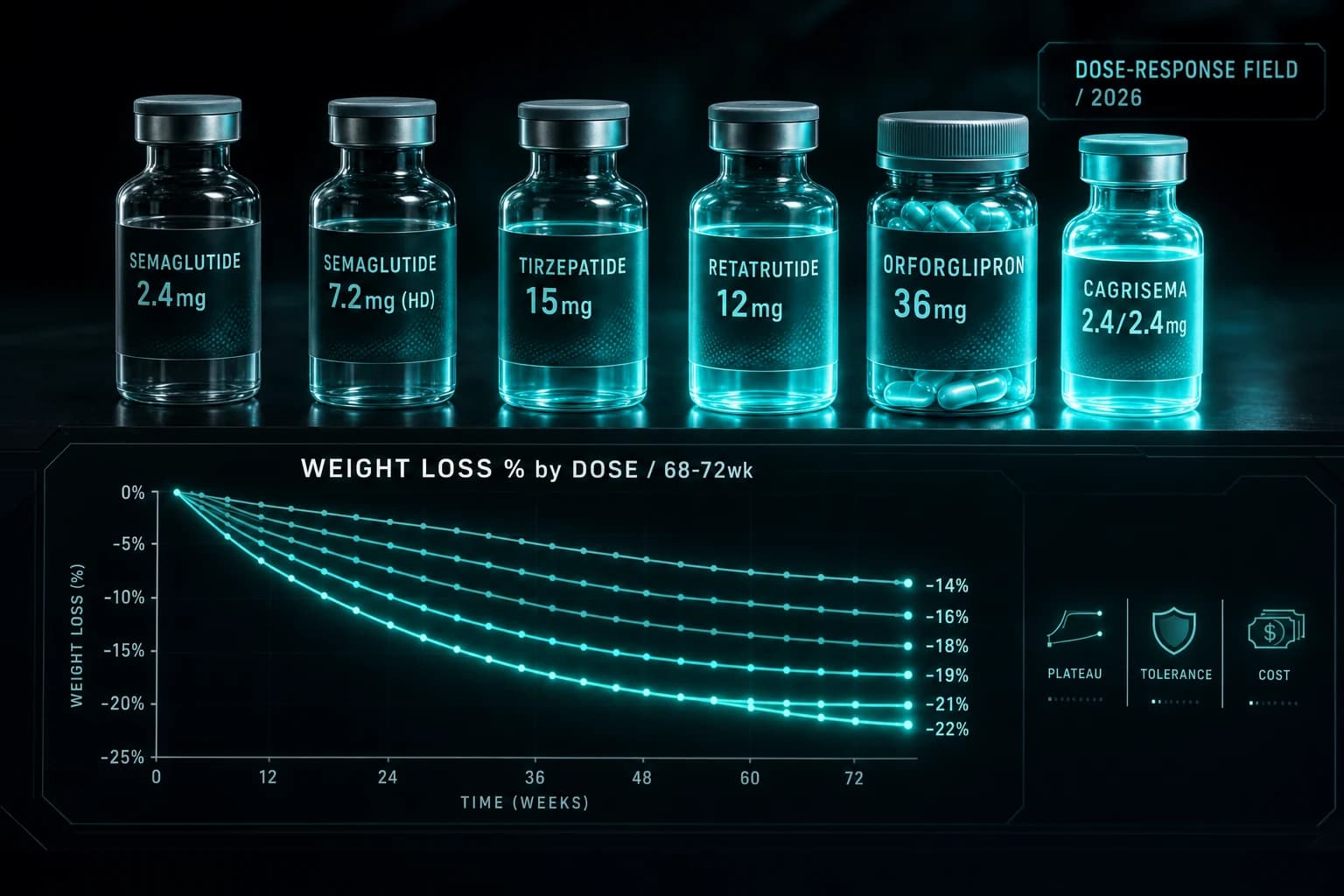
- Wegovy HD 7.2 mg was FDA approved March 19, 2026 with 20.7% mean weight loss at 72 weeks in STEP UP, the highest result for any semaglutide formulation to date
- Tirzepatide 15 mg in SURMOUNT-1 produced 22.5% weight loss at 72 weeks, still the highest result among injectable approved GLP-1 class drugs
- Retatrutide 12 mg in Phase 2 produced 24.2% weight loss at 48 weeks and is still climbing at the top dose, with no clear plateau yet
- Orforglipron 36 mg oral produced about 12% weight loss at 72 weeks in ATTAIN-1, less than the injectable class but with no needles
- Dose escalation is mandatory across the class; jumping from a 2.4 mg standard dose to a 7.2 mg high-dose without titration is not how Wegovy HD or any other agent in this class works
The GLP-1 dosing question changed in March 2026. The FDA approved Wegovy HD (semaglutide 7.2 mg) on March 19, 2026, with 20.7% mean weight loss at 72 weeks in the STEP UP trial. That number puts semaglutide on the same dose-response shelf as tirzepatide 15 mg for the first time since the field opened up, and it raises the central question every researcher and patient is now asking: what is the right dose, when do you escalate, and where does the dose-response curve actually plateau?
This article maps the complete 2026 dose landscape across the GLP-1 class. Semaglutide from 0.25 mg starter through 7.2 mg high-dose. Tirzepatide from 2.5 mg through 15 mg. Retatrutide Phase 2 doses through 12 mg. Orforglipron oral doses. Combination dosing for CagriSema, MariTide, mazdutide. What the published dose-response curves actually show, where the diminishing returns kick in, and how to think about picking a research or therapeutic dose when there are now six different active doses of semaglutide alone.
What changed in 2026
Three things shifted the dosing conversation this year.
First, Wegovy HD 7.2 mg was approved under the FDA Commissioner's National Priority Voucher pilot program on March 19, 2026, becoming the first semaglutide expansion above 2.4 mg (Novo Nordisk press release, 2026). The STEP UP Phase 3 trial in approximately 1,400 adults with obesity produced 20.7% mean weight loss at 72 weeks, and 31.2% of participants reached at least 25% weight loss. In the type 2 diabetes arm, the same 7.2 mg dose produced 14.1% weight loss, which is the highest semaglutide result in the diabetes population to date.
Second, the retatrutide Phase 2 data at 12 mg has held up to longer-term analysis with 24.2% weight loss at 48 weeks and no clear plateau at the top dose (Jastreboff et al., NEJM, 2023). Phase 3 is ongoing.
Third, orforglipron gave the oral GLP-1 class a real entry. The ATTAIN-1 Phase 3 trial produced about 12% weight loss at 72 weeks at the 36 mg dose, less than the injectable class but enough to make oral GLP-1 a real lever in the discussion.
The cumulative effect is that the GLP-1 dose-response question is no longer about whether to dose 1.7 mg or 2.4 mg semaglutide. It is now: where on a 6-or-more step dose ladder, across at least 5 different active molecules, is the right place to land for a given goal?
The complete 2026 dose-response table
| Compound | Brand | Dose ladder | Trial | Best dose | Mean weight loss | Duration | Status |
|---|---|---|---|---|---|---|---|
| Semaglutide | Wegovy | 0.25 → 0.5 → 1.0 → 1.7 → 2.4 mg | STEP 1 | 2.4 mg | -14.9% | 68 wk | FDA approved |
| Semaglutide HD | Wegovy HD | 0.25 → 0.5 → 1.0 → 1.7 → 2.4 → 4.8 → 7.2 mg | STEP UP | 7.2 mg | -20.7% | 72 wk | FDA approved (March 2026) |
| Tirzepatide | Zepbound | 2.5 → 5 → 7.5 → 10 → 12.5 → 15 mg | SURMOUNT-1 | 15 mg | -22.5% | 72 wk | FDA approved |
| Retatrutide | (none) | 0.5 → 2 → 4 → 6 → 8 → 12 mg | Phase 2 | 12 mg | -24.2% | 48 wk | Phase 3 ongoing |
| Orforglipron (oral) | (none) | 1 → 3 → 6 → 12 → 24 → 36 mg | ATTAIN-1 | 36 mg | ~12% | 72 wk | Filed |
| CagriSema | (none) | 0.25/0.25 → 2.4/2.4 mg | REDEFINE-1 | 2.4/2.4 mg | -22.7% | 68 wk | Filed |
| Survodutide | (none) | 0.6 → 4.8 mg | Phase 2 | 4.8 mg | -16.6% on-treatment | 46 wk | Phase 3 ongoing |
| Mazdutide | (none) | 4.5 → 6 → 9 mg | GLORY-1 | 9 mg | ~15% | 48 wk | Phase 3 |
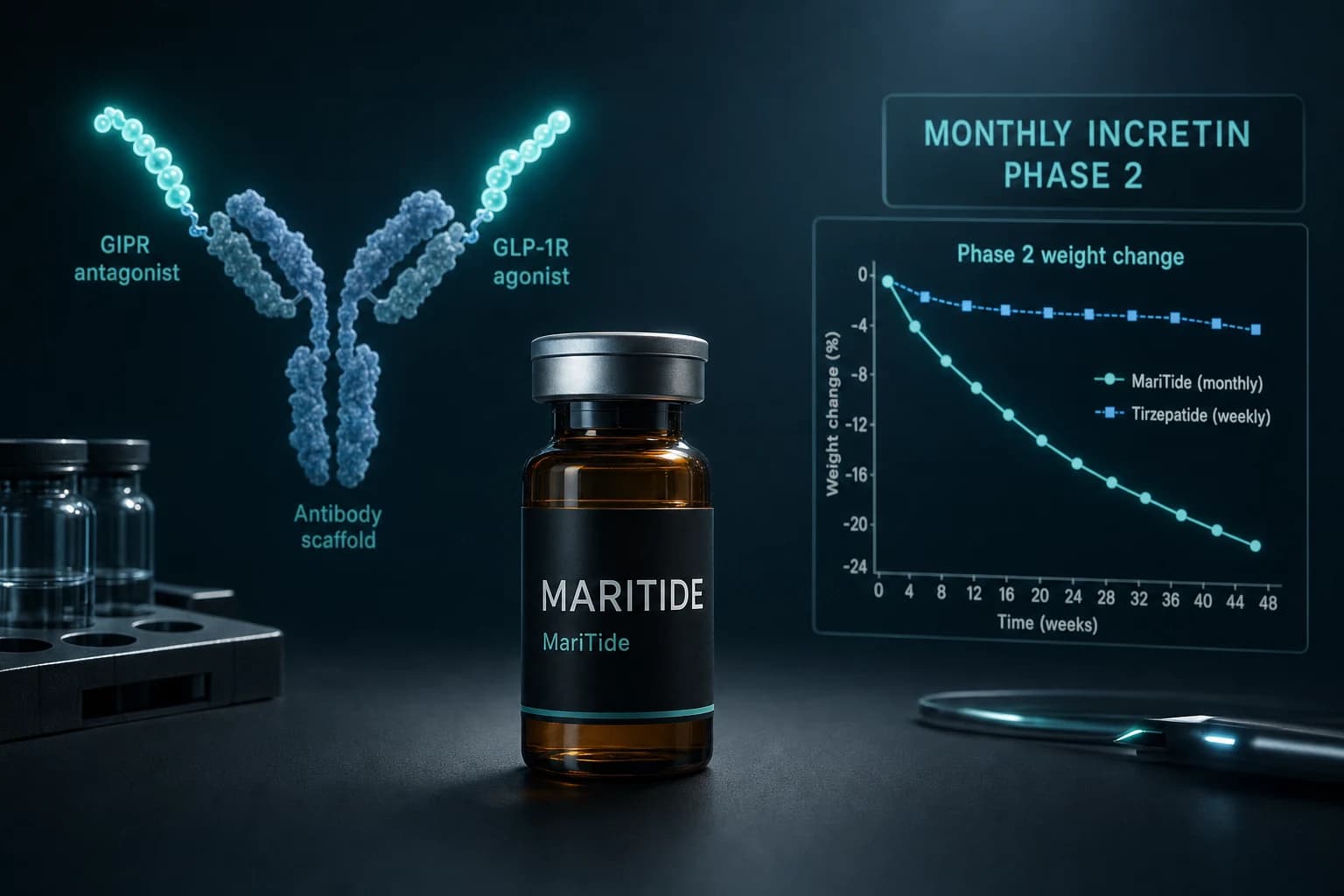
| MariTide | (none) | varying | Phase 2 | top dose | ~20% | 52 wk | Phase 3 starting |
A few things jump out before zooming in compound-by-compound.
Tirzepatide still has the highest approved-drug weight loss number. Wegovy HD closed most of the gap (20.7% vs 22.5%) but tirzepatide 15 mg from SURMOUNT-1 remains the published ceiling among FDA-approved agents.
Retatrutide is the highest weight-loss number, period. 24.2% at 48 weeks in Phase 2. Whether Phase 3 confirms this is the central pipeline question of 2026.
Combination dosing operates on different math. CagriSema's 22.7% comes from running 2.4 mg semaglutide alongside 2.4 mg cagrilintide. That is a mechanism-stacked dose, not a higher-dose monotherapy result.
Oral GLP-1 has its own dose ladder. Orforglipron 36 mg at 12% is not directly comparable to injectable doses because the route and bioavailability are different. The oral class is its own dose conversation.
Bottom line: Tirzepatide 15 mg holds the highest approved-drug weight loss number (22.5%). Wegovy HD 7.2 mg closed most of the gap (20.7%) but did not surpass tirzepatide. Retatrutide 12 mg leads at 24.2% but is still Phase 3. Combination therapy (CagriSema) and multi-receptor monotherapy (tirzepatide, retatrutide) both produce more weight loss than mono-GLP-1 at the same dose tier.
Semaglutide doses: 0.25 → 2.4 → 7.2 mg
Standard Wegovy uses a 5-step dose ladder over 17 weeks before reaching the 2.4 mg target. The full pen sequence is 0.25, 0.5, 1.0, 1.7, 2.4 mg, each held for 4 weeks. This ramp exists because GI side effects (nausea, constipation, vomiting) scale with dose, and the slow titration lets gut adaptation keep pace.
The STEP 1 trial (Wilding et al., NEJM, 2021) randomized 1,961 adults with obesity to weekly semaglutide 2.4 mg or placebo, plus lifestyle intervention, for 68 weeks. Result: 14.9% mean weight loss with semaglutide versus 2.4% with placebo, an 11.6 percentage point treatment effect at the 2.4 mg dose.
Wegovy HD adds two more steps to the ladder: 4.8 mg held for 4 weeks, then 7.2 mg as the maintenance dose. The full titration to 7.2 mg now runs roughly 25 weeks before reaching maintenance.
The STEP UP trial that supported the HD approval used this longer ladder. Approximately 1,400 adults with obesity were randomized to semaglutide 7.2 mg, semaglutide 2.4 mg, or placebo for 72 weeks. Result: 20.7% mean weight loss at 7.2 mg, with 31.2% of participants reaching ≥25% weight loss. This is the highest published weight loss result for any semaglutide formulation.
The dose-response shape from STEP 1 (2.4 mg) to STEP UP (7.2 mg) shows continued response, not plateau. Tripling the dose produced approximately a 6 percentage point additional weight loss at matched timepoints, suggesting the semaglutide dose-response curve had not flattened at 2.4 mg.
Side effects also scaled. GI side effect rates were higher at 7.2 mg than at 2.4 mg in STEP UP, but the discontinuation rate due to GI events did not increase dramatically, partly because the longer titration gives gut adaptation more time. The HD label specifies adults with obesity who tolerated the 2.4 mg dose first, not direct initiation at the higher dose.
For our deeper coverage of semaglutide reconstitution and dosing math, see the semaglutide reconstitution chart. For the semaglutide compound research overview, see the semaglutide compound guide.
Tirzepatide doses: 2.5 → 15 mg
Tirzepatide uses a 6-step dose ladder: 2.5, 5, 7.5, 10, 12.5, 15 mg, each held for 4 weeks. Full titration to 15 mg takes 20 weeks.
The SURMOUNT-1 trial (Jastreboff et al., NEJM, 2022) randomized 2,539 adults with obesity to placebo, tirzepatide 5 mg, 10 mg, or 15 mg for 72 weeks. Results:
| Dose | Mean weight loss at 72 wk |
|---|---|
| Placebo | -3.1% |
| Tirzepatide 5 mg | -15.0% |
| Tirzepatide 10 mg | -19.5% |
| Tirzepatide 15 mg | -20.9% |
The dose-response from 5 to 15 mg shows steeper gains between 5 and 10 mg than between 10 and 15 mg. The 4.5 percentage point gain from 5 → 10 mg compresses to a 1.4 percentage point gain from 10 → 15 mg. This is the classic shape of a curve approaching a plateau, but not necessarily fully plateaued. The SURMOUNT-1 estimand using on-treatment data put 15 mg at 22.5%.
The practical implication: for most patients, 10 mg captures most of the available weight loss, and the climb to 15 mg adds incremental benefit at increased side effect burden. The 15 mg dose is the right answer when 10 mg has not produced sufficient response or when the patient tolerates the higher dose well.
For our deeper coverage of tirzepatide dosing, see the tirzepatide reconstitution chart. For the head-to-head against semaglutide, see tirzepatide vs semaglutide 2026.
Retatrutide doses: 0.5 → 12 mg
Retatrutide is the triple agonist (GIP + GLP-1 + glucagon) currently in Phase 3. The published dose-response is from the Phase 2 trial (Jastreboff et al., NEJM, 2023) which randomized 338 adults with obesity to retatrutide 1, 4, 8, 12 mg, or placebo for 48 weeks.
| Dose | Mean weight loss at 48 wk |
|---|---|
| Placebo | -2.1% |
| Retatrutide 1 mg | -8.7% |
| Retatrutide 4 mg | -17.1% |
| Retatrutide 8 mg | -22.8% |
| Retatrutide 12 mg | -24.2% |
What makes this curve different from tirzepatide and semaglutide is that the top-dose result of 24.2% at 48 weeks is meaningfully higher than tirzepatide's 22.5% at 72 weeks. Tirzepatide had 24 more weeks of treatment time to reach that number. The retatrutide curve at 48 weeks suggests it would still be climbing at 72 weeks if the trial had run longer.
The dose escalation in the Phase 2 trial used 4-week steps roughly doubling the dose: 0.5, 1, 2, 4, 6, 8, 12 mg. The 12-week retatrutide titration protocol covers the practical research-grade dose ramp in detail.
For comparison-class context, see retatrutide vs tirzepatide vs semaglutide.
Orforglipron doses: 6 → 36 mg (oral)
Orforglipron is the Lilly oral GLP-1 receptor agonist filed in late 2025. The Phase 3 ATTAIN program tested 1, 3, 6, 12, 24, and 36 mg once-daily oral doses.
ATTAIN-1 in adults with obesity produced approximately 12.4% mean weight loss at 36 mg over 72 weeks. The dose-response was relatively flat from 24 to 36 mg, suggesting the curve was approaching saturation at the upper doses.
The dose ladder for orforglipron uses 4-week titration steps similar to the injectable class. Standard escalation is 1, 3, 6, 12, 24, 36 mg, each held for 4 weeks.
The key dosing distinction for orforglipron is the route. Oral bioavailability is lower than subcutaneous injection, which is why the milligram doses are an order of magnitude higher than the injectable comparators. Comparing the 36 mg oral dose to a 2.4 mg injectable dose by milligram is the wrong axis. The right axis is the weight loss endpoint, where the oral dose produces a smaller effect than injectable semaglutide 2.4 mg.
For deeper coverage of orforglipron, see the orforglipron Phase 3 evidence article.
CagriSema dosing: 2.4/2.4 mg combination
CagriSema is the fixed-dose combination of cagrilintide (long-acting amylin analog) plus semaglutide, filed by Novo Nordisk in late 2025. The dose is reported as 2.4/2.4 mg, meaning 2.4 mg of each agent per weekly injection.
The REDEFINE-1 Phase 3 trial produced 22.7% mean weight loss at 68 weeks at the 2.4/2.4 mg target dose. This is the highest published result for a fixed-dose obesity combination and one of the highest results in the entire weight-loss class.
The dose ladder for CagriSema mirrors standard semaglutide titration: 0.25/0.25, 0.5/0.5, 1.0/1.0, 1.7/1.7, 2.4/2.4 mg, each held for 4 weeks. The amylin component scales in lockstep with the GLP-1 component.
The mechanistic logic: amylin and GLP-1 work through complementary satiety pathways. The combination dose produces additive effects at less than 2x the dose escalation. Running 2.4 mg cagrilintide alongside 2.4 mg semaglutide produces more weight loss than running 7.2 mg semaglutide alone (22.7% vs 20.7%), but the trials were different populations and durations so the comparison is approximate.
For deeper coverage, see the CagriSema REDEFINE Phase 3 guide.
Other Phase 3 doses: survodutide, mazdutide, MariTide
The pipeline has three more GLP-1-class agents at meaningful dose ladders.
Survodutide is the Boehringer/Zealand GCG/GLP-1 dual agonist. Phase 2 used 0.6, 2.4, 3.6, and 4.8 mg weekly. The 4.8 mg dose produced 16.6% on-treatment weight loss at 46 weeks. The survodutide Phase 2 evidence covers the trial design and how the GCG mechanism behaves at the top dose.
Mazdutide is the Innovent/Lilly GLP-1/glucagon dual agonist with Chinese Phase 3 data. The dose ladder is 4.5, 6, 9 mg weekly. The 9 mg dose produced approximately 15% weight loss at 48 weeks in GLORY-1. The mazdutide Phase 3 evidence covers the published data.
MariTide (maridebart cafraglutide) is the Amgen GIP-antagonist / GLP-1 agonist hybrid. Phase 2 produced approximately 20% weight loss at 52 weeks at the top dose. The dose ladder is monthly rather than weekly, which is a structural distinction worth understanding before extrapolating from the weekly class. The MariTide Phase 2 evidence covers the unique mechanism and dosing schedule.
What the dose-response curves actually show
Across the full class, the dose-response curves share three patterns:
Pattern 1: Steep gain in the low-dose range, flatter in the mid-range. Going from placebo to the first active dose typically produces 6-10 percentage points of weight loss. Each subsequent dose step adds less. This is the classic concave curve approaching an asymptote.
Pattern 2: The asymptote is higher for multi-receptor compounds. Tirzepatide (dual GIP/GLP-1) plateaus higher than semaglutide (mono-GLP-1). Retatrutide (triple GIP/GLP-1/glucagon) climbs higher than tirzepatide at matched durations. The number of receptors a compound hits sets the eventual ceiling.
Pattern 3: Side effect curves rise more linearly than efficacy curves. This is the dose-finding problem. The weight-loss benefit per mg flattens. The nausea/vomiting rate per mg does not flatten the same way. Beyond some dose, the marginal benefit shrinks while the marginal harm continues.
For most patients on most agents, the practical sweet spot lands one or two steps below the top of the ladder. Tirzepatide 10 mg captures most of the available effect. Semaglutide 2.4 mg captures most of the standard-Wegovy effect. Semaglutide 7.2 mg unlocks a meaningful new tier but at additional side effect burden. Retatrutide is the exception so far in that the curve has not clearly turned over by 12 mg.
Compounded vs branded high-dose
The compounded peptide market has been selling research-grade semaglutide and tirzepatide at doses outside the branded ladder for years. With the Wegovy HD approval, the gap between branded high-dose and research-grade dosing has narrowed.
A few practical distinctions:
-
Branded titration follows a tested ladder. Wegovy HD's 7.2 mg dose is reached after 25 weeks of titration through 4.8 mg. A compounded 7.2 mg vial used without titration is not a research-validated protocol; it is a self-built dose escalation that may produce significantly worse tolerance.
-
Compounded doses can be tailored. Research-grade vials in 5 mg, 10 mg, and 30 mg sizes allow precise dose-by-volume titration that the branded pen does not. For researchers running comparative work or tight titration, this is a meaningful flexibility.
-
The FDA 503A compounding picture is changing. The July 2026 PCAC vote on the 503A peptide bulks list will influence which compounded peptides remain legally available. The FDA 503A peptide compounding review covers the regulatory state.
For dosing precision, the reconstitution calculator handles arbitrary vial sizes and target doses. The semaglutide dosage chart, tirzepatide dosage chart, and retatrutide dosage chart cover compound-specific math.
How to think about picking a dose
For a researcher or patient choosing where to land on a GLP-1 dose ladder, four questions usually frame the choice.
1. What is the weight-loss goal? A 10-15% target lands in the standard-dose range for most compounds (semaglutide 2.4 mg, tirzepatide 5-10 mg, mazdutide 6 mg). A 15-22% target requires the higher end (semaglutide 7.2 mg, tirzepatide 10-15 mg, retatrutide 8 mg, CagriSema 2.4/2.4 mg). A 22+% target currently requires retatrutide 12 mg, CagriSema 2.4/2.4 mg, or tirzepatide 15 mg with longer durations.
2. What does tolerance look like at the previous step? If 2.4 mg semaglutide is producing tolerable side effects, the step to 4.8 → 7.2 mg has a reasonable risk profile. If 2.4 mg already produces significant nausea, 7.2 mg is not the right escalation. Tolerance at the standard dose is the gating signal.
3. What is the cost and access profile? Branded high-dose pens cost more than standard-dose pens. Insurance coverage and supply chain availability differ across compounds and doses. The cost-per-percentage-point-of-weight-loss math changes meaningfully at higher doses where the curve is flatter.
4. What is the maintenance plan? Stopping at the top dose tends to produce more weight regain than stopping at a moderate dose, partly because the higher dose was carrying more of the satiety lift that disappears on discontinuation. The stopping GLP-1 and weight regain research covers what happens after discontinuation across the class.
The right dose is rarely the highest dose. It is the dose where the marginal benefit still justifies the marginal harm for the specific patient or research goal.
Bottom line: For most patients, one or two steps below the top of the ladder captures the bulk of the available weight loss without the disproportionate side effect burden. Tirzepatide 10 mg, semaglutide 2.4 mg, retatrutide 8 mg are the practical sweet spots. The top dose is worth pursuing when tolerance is good and the goal has not been reached.
Sourcing notes
For research-grade injectable semaglutide, tirzepatide, retatrutide, and cagrilintide vials with public per-batch COAs, Ascension Peptides ships with 50% off using code ENHANCED. For oral metabolic compounds including tesofensine and 5-amino-1MQ as adjacent comparison work, Limitless Biotech is the partner for oral formats with code ENHANCED.
For our deeper sourcing analysis, see the best legit peptide vendors 2026 ranking. For specific buyer-intent guides, see the retatrutide where-to-buy and retatrutide reconstitution coverage.
FAQ
Should I jump from semaglutide 2.4 mg straight to 7.2 mg?
No. Wegovy HD is approved with a titration through 4.8 mg, held for 4 weeks before escalating to 7.2 mg. The full ramp from 2.4 mg to 7.2 mg adds approximately 8 weeks to the existing titration. Jumping from 2.4 mg directly to 7.2 mg without the intermediate step is not the approved protocol and will produce materially worse GI tolerance.
Is the 7.2 mg dose better than tirzepatide 15 mg?
Tirzepatide 15 mg produced 22.5% weight loss at 72 weeks in SURMOUNT-1. Semaglutide 7.2 mg produced 20.7% at 72 weeks in STEP UP. Tirzepatide remains the higher number among approved drugs, but the gap is now under 2 percentage points. Direct head-to-head head trial data at these doses is not yet published. For most patients, the choice depends on individual tolerance and access more than the on-paper difference.
What does the retatrutide 12 mg result mean for the field?
Retatrutide 12 mg produced 24.2% weight loss at 48 weeks in Phase 2, the highest published result in the obesity class. Phase 3 is ongoing. If Phase 3 confirms the Phase 2 numbers, retatrutide will establish a new ceiling for monotherapy weight loss above what any GIP/GLP-1 or mono-GLP-1 agent has produced. The triple-receptor mechanism (GIP + GLP-1 + glucagon) is the structural reason.
Why is the oral orforglipron dose so much higher (36 mg) than the injectable doses?
Oral bioavailability is lower than subcutaneous injection. The 36 mg oral dose produces systemic exposure roughly comparable to or below what 2.4 mg injectable semaglutide produces. Comparing milligrams across routes is the wrong axis. The right axis is the weight-loss endpoint, and on that axis the orforglipron 36 mg oral result is meaningfully below the injectable class.
What about compounded semaglutide above 2.4 mg?
Research-grade vials at 5, 10, and 30 mg sizes have been available through peptide vendors for years, and researchers have been running doses above 2.4 mg before Wegovy HD was approved. The Wegovy HD approval validates the dose-response concept above 2.4 mg with a Phase 3 trial. The compounded market predates that approval and has been running similar doses without the titration data. The key practical distinction is the titration ladder: Wegovy HD reaches 7.2 mg after a tested 25-week ramp. A self-built ramp may be longer, shorter, or different, and tolerance varies accordingly.
Does CagriSema's 22.7% mean the combination is better than monotherapy?
The combination of cagrilintide and semaglutide hits two separate satiety pathways (amylin and GLP-1), and the 22.7% result at 2.4/2.4 mg is higher than 2.4 mg semaglutide alone (14.9%) and higher than 7.2 mg semaglutide alone (20.7%). The combination is mechanistically distinct from a higher monotherapy dose. For the same total weekly dose, the combination produces more weight loss than running semaglutide alone at any dose currently tested.
When does dose escalation become diminishing returns?
For tirzepatide, the gain from 10 → 15 mg was 1.4 percentage points over 5 percentage points from 5 → 10 mg. That is a clear diminishing-returns signal. For semaglutide, the gain from 2.4 → 7.2 mg was approximately 6 percentage points over 11 percentage points from 0 → 2.4 mg, also slowing but less steeply. For retatrutide, the gain from 8 → 12 mg was 1.4 percentage points, also showing flattening at the top. Across the class, the curve flattens beyond the "second-from-top" dose for most compounds. The practical implication is that the top dose is usually right for patients who tolerate it well and have not reached their goal, but the gain over the second-from-top dose is typically modest.
What is the right starting dose for research?
For semaglutide research, 0.25 mg weekly is the standard 4-week starting dose. For tirzepatide research, 2.5 mg weekly. For retatrutide research, 0.5 mg weekly. Each agent has a tested ramp protocol that should be respected for tolerance reasons. The reconstitution calculator handles dose-by-volume math for arbitrary vial sizes.
Further reading
- Tirzepatide vs Semaglutide 2026 head-to-head
- Retatrutide vs Tirzepatide vs Semaglutide
- CagriSema REDEFINE Phase 3 guide
- Orforglipron Phase 3 evidence
- Survodutide Phase 2 evidence
- Mazdutide Phase 3 evidence
- MariTide Phase 2 evidence
- Retatrutide 12-week titration protocol
- Tirzepatide reconstitution chart 10mg & 15mg
- Semaglutide reconstitution chart 5mg
- Retatrutide reconstitution guide 30mg
- GLP-1 muscle loss research
- Stopping GLP-1 and weight regain
- Reconstitution Calculator
This article is for educational and research purposes only. Semaglutide is FDA approved as Wegovy and Wegovy HD for chronic weight management and as Ozempic for type 2 diabetes. Tirzepatide is FDA approved as Zepbound for chronic weight management and obstructive sleep apnea, and as Mounjaro for type 2 diabetes. Retatrutide, survodutide, mazdutide, MariTide, orforglipron, and cagrilintide-semaglutide (CagriSema) are not FDA approved as of May 2026. None of the content above constitutes medical advice. Patients managing obesity or type 2 diabetes should work with qualified clinicians on appropriate dose selection and titration; nothing in this article replaces that.