At a glance
- PCAC meets July 23-24, 2026 to vote on adding 7 peptides to the 503A bulks list
- Day 1: BPC-157 (UC), KPV (wound healing), TB-500 (wound healing), MOTS-c (obesity, osteoporosis)
- Day 2: DSIP/Emideltide (insomnia, opioid withdrawal, narcolepsy), Semax (cerebral ischemia, migraine), Epitalon
- All 7 came off the Category 2 'significant safety risks' list on April 22, 2026
- PCAC votes are advisory; formal 503A inclusion still requires notice-and-comment rulemaking
- Public comment docket FDA-2025-N-6895 closes July 22, 2026
What's actually being decided
On July 23 and 24, 2026, the FDA's Pharmacy Compounding Advisory Committee (PCAC) will hold a two-day meeting to vote on whether seven peptides should be added to the Section 503A Bulk Drug Substances list. If a peptide makes that list, licensed compounding pharmacies can produce it from bulk under specified terms. If it does not, compounding becomes legally tenuous and, in practice, often stops.
The seven peptides are split across the two days:
- Day 1 (July 23): BPC-157, KPV, TB-500, and MOTS-c
- Day 2 (July 24): Emideltide (also called DSIP), Semax, and Epitalon
Each substance has a proposed clinical use that PCAC will weigh against the published evidence. The public comment docket is FDA-2025-N-6895, open until July 22, 2026.
Note: PCAC votes are advisory only. Even a unanimous "yes" still requires the FDA to publish a proposed rule, take public comment, and issue a final rule before a peptide is formally on the 503A list.
Why this meeting exists at all
Compounded drugs sit in a regulatory carve-out. Section 503A of the Federal Food, Drug, and Cosmetic Act lets state-licensed pharmacies compound prescriptions for specific patients without going through new-drug approval, but only if the bulk active ingredient is one of three things: a USP/NF monograph compound, a component of an FDA-approved drug, or on the 503A Bulks List.
Peptides like BPC-157 and TB-500 have never been any of those. They sat for years in a regulatory grey zone the FDA classified as "Category 2," meaning the agency had identified significant safety concerns and considered them ineligible for compounding. That category was non-binding interim guidance, but most compounding pharmacies treated it as a hard line.
In April 2026, FDA reclassified twelve peptides off the Category 2 list. The July PCAC meeting is the next formal step: a recommendation on whether seven of them should be officially added to the 503A bulks list, with specific allowed uses.
Three months out from the vote, the evidence picture varies a lot by compound.
Day 1: BPC-157, KPV, TB-500, MOTS-c
| Compound | Proposed use | Strongest published evidence | Evidence tier |
|---|---|---|---|
| BPC-157 | Ulcerative colitis | Phase 2 trials (PL-10/PL14736) and rat colitis models | Animal plus small unpublished Phase 2 |
| KPV | Wound healing, inflammatory conditions | DSS and TNBS murine colitis (Dalmasso 2008) | Preclinical |
| TB-500 | Wound healing | Phase 2 venous stasis and pressure ulcer trials | Small Phase 2 in humans |
| MOTS-c | Obesity, osteoporosis | Diet-induced obesity in mice (Lee 2015) | Preclinical |
BPC-157 for ulcerative colitis
BPC-157 is the most-cited peptide of the seven, with hundreds of preclinical papers and a long thread of work led by Predrag Sikiric at the University of Zagreb. The colitis case rests on rat models of cysteamine, DSS, and ischemia-reperfusion colitis where BPC-157 spared mucosa and reduced ulceration (Sikiric et al. World J Gastroenterol 2017).
Some Phase 2 work in inflammatory bowel disease was reported by Pliva (Croatia) under names like PL-10 and PL14736, but the trials were small, never published as full peer-reviewed reports, and never advanced to Phase 3.
Forum and clinical use of BPC-157 mostly target tendon, ligament, and gut mucosal recovery rather than ulcerative colitis specifically. The "UC" framing is a sponsor decision: it gives PCAC a clinical indication where the rodent evidence is densest, even though that is not what most current researchers actually use it for.
Bottom line: BPC-157 has the deepest preclinical literature of the seven, but human evidence is limited. Approval for UC would be a narrow lane; everything else stays off-label.
KPV for wound healing and inflammation
KPV is the C-terminal three amino acids of alpha-MSH (lysine, proline, valine). The anti-inflammatory case is mechanistically clean: KPV inhibits NF-kappaB and IL-1beta signaling in intestinal epithelium and immune cells, and oral KPV reduced inflammation in DSS- and TNBS-induced murine colitis (Dalmasso et al. Gastroenterology 2008).
Human clinical evidence is the weakest of the four Day-1 peptides. There are no published randomized human trials. The proposed use ("wound healing and inflammatory conditions") is broader than the published evidence supports: most positive data is colitis-specific, with much smaller datasets in skin and oral inflammation.
Compounding-pharmacy use cases are mostly oral capsules for gut inflammation and topical formulations for skin and oral conditions, not the same mucosal indication BPC-157 occupies.
TB-500 for wound healing
TB-500 is a synthetic version of an actin-binding portion of thymosin beta-4. Two Phase 2 trials in venous stasis and pressure ulcers reported accelerated healing in patients who responded (Treadwell, Kleinman, Crockford, Hardy, Guarnera, Goldstein. Ann N Y Acad Sci 2012).
The evidence base for TB-500 is the most clinically anchored of the Day-1 peptides for wound healing specifically. The Phase 2 work was sponsored by RegeneRx; subsequent Phase 3 development has been intermittent.
Most off-label use of TB-500 is in soft-tissue injury recovery rather than the chronic ulcer populations the trials enrolled. PCAC will likely focus on whether the proposed use ("wound healing") is too broad relative to the studied population.
For a deeper comparison of BPC-157 and TB-500 in recovery contexts, see the Wolverine Stack protocol and the BPC-157 vs TB-500 guide.
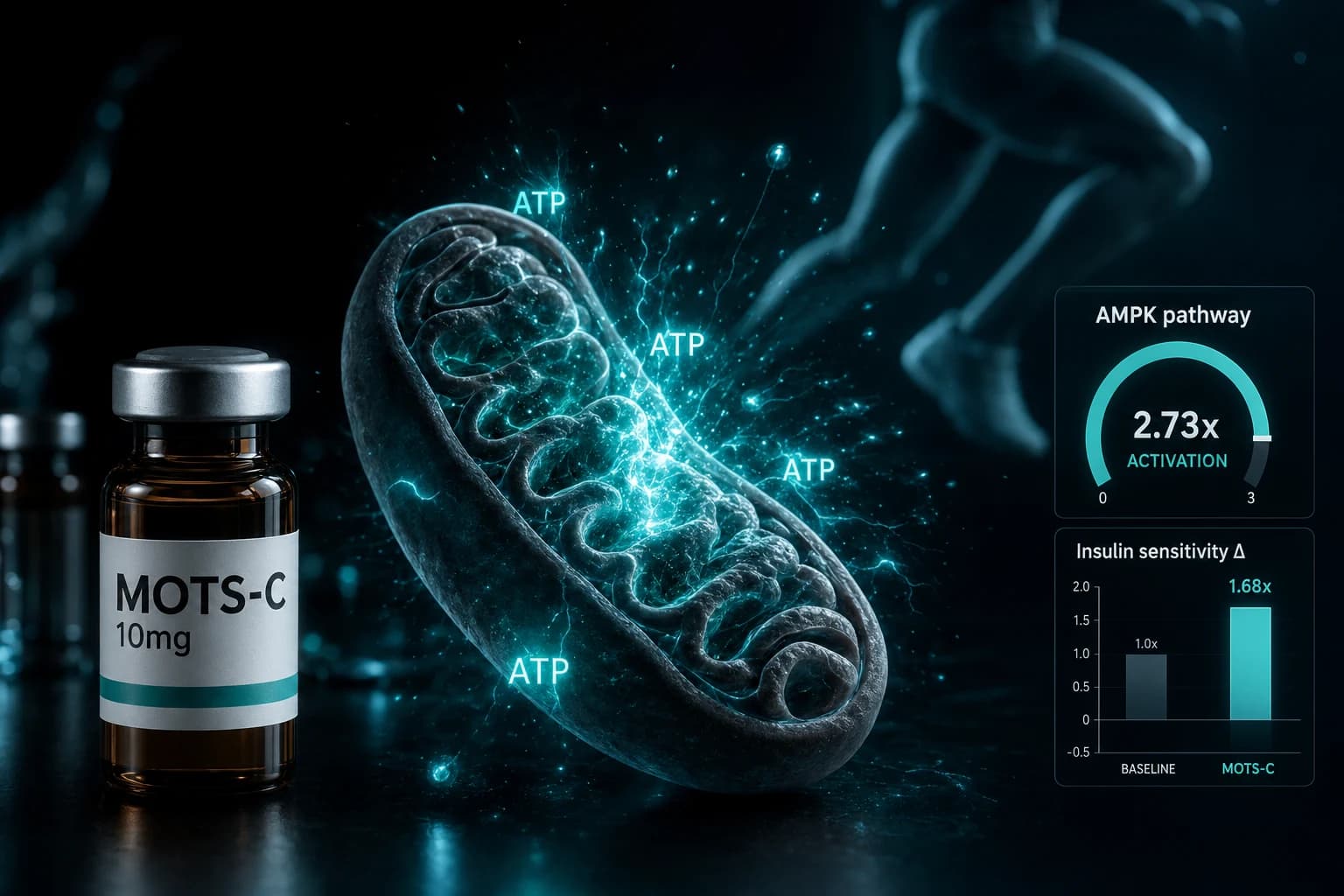
MOTS-c for obesity and osteoporosis
MOTS-c is a 16-amino-acid mitochondrial-derived peptide with strong preclinical metabolic data. It improves insulin sensitivity, reduces diet-induced obesity in mice, and activates AMPK in skeletal muscle (Lee et al. Cell Metab 2015).
The osteoporosis claim is much thinner. There are scattered preclinical papers on bone, but no clinical osteoporosis trials. Combining "obesity and osteoporosis" in one nomination is unusual and may draw scrutiny. PCAC reviewers tend to prefer narrowly defined uses with a clear study population.
For more on MOTS-c mechanism and exercise mimetic data, see the MOTS-c research guide.
Day 2: DSIP, Semax, Epitalon
| Compound | Proposed use | Strongest published evidence | Evidence tier |
|---|---|---|---|
| Emideltide / DSIP | Opioid withdrawal, chronic insomnia, narcolepsy | Small human sleep trials in insomniacs (1981) | Old, small human |
| Semax | Cerebral ischemia, migraine, trigeminal neuralgia | Russian stroke and BDNF studies | Russian clinical, mostly untranslated |
| Epitalon | Specific use to be clarified in docket | Telomerase activation in human cell lines | Cell culture plus Russian gerontology |
DSIP / Emideltide
DSIP is a nine-amino-acid peptide isolated in the 1970s by the Schoenenberger-Monnier group from sleeping rabbit cerebral venous blood. The supporting human data is older and small: synthetic DSIP at 25 nmol/kg IV in 6 chronic insomniacs improved sleep duration and quality without next-day sedation (Schneider-Helmert. Experientia 1981).
The proposed uses (opioid withdrawal, chronic insomnia, narcolepsy) are broader than what those trials tested. The trials are also old enough that PCAC reviewers will likely raise modern methodology concerns: small n, no proper placebo control by current standards, and no replication in larger cohorts.
For nootropic and sleep-stack context, see the Calm and Clarity stack, which covers adjacent compounds without depending on DSIP specifically.
Semax
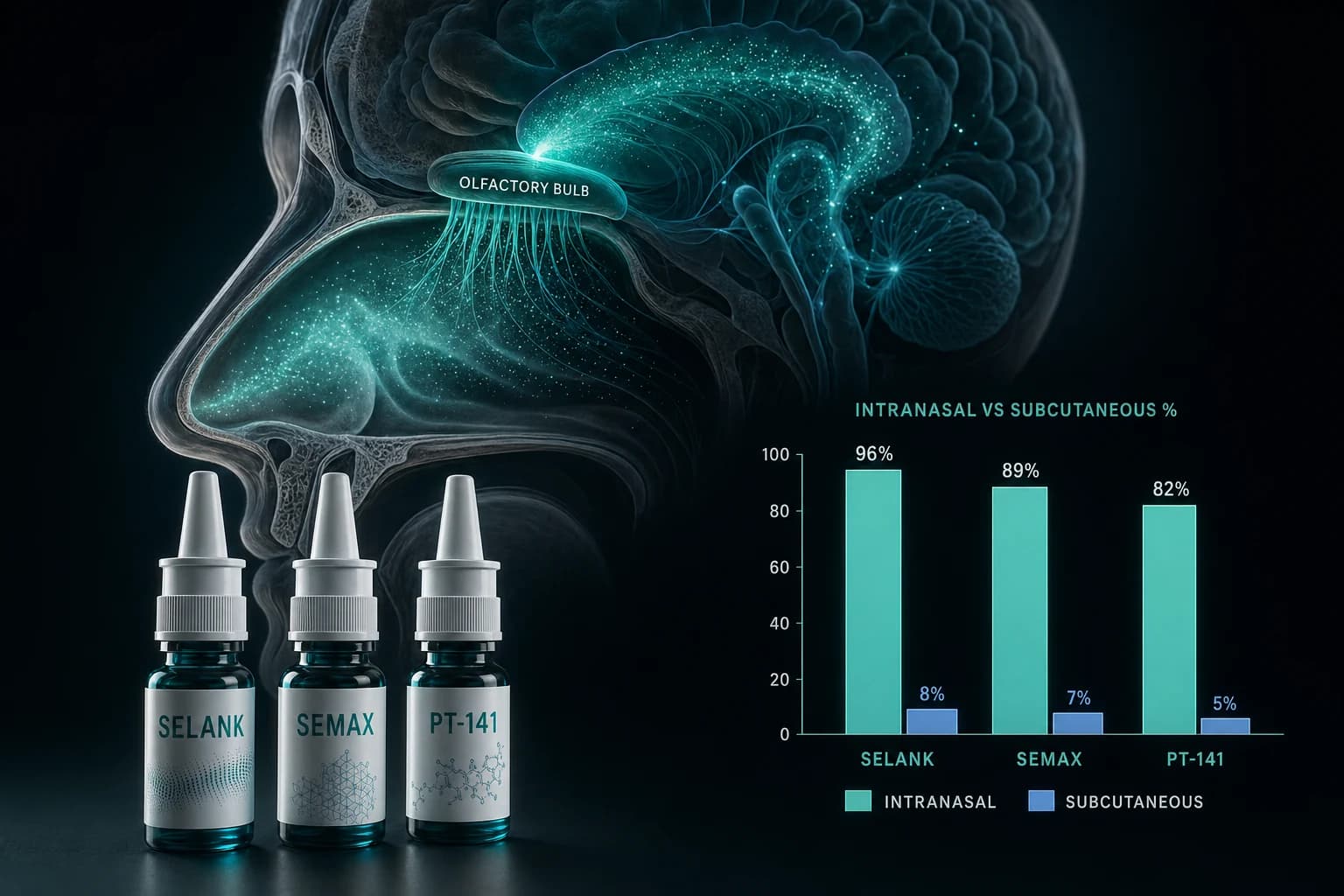
Semax is a heptapeptide analog of ACTH(4-10), developed in Russia and on the Russian List of Vital and Essential Drugs since 2011. Mechanistic work in rats shows rapid BDNF and TrkB upregulation in the hippocampus after intranasal dosing (Dolotov et al. 2006).
The proposed uses (cerebral ischemia, migraine, trigeminal neuralgia) align with how Semax is actually used in Russian clinical practice. The challenge for PCAC is that most of the supporting clinical literature is in Russian-language journals, has not been replicated in Western randomized controlled trials, and may not meet modern methodological standards.
Epitalon
Epitalon is a synthetic tetrapeptide (Ala-Glu-Asp-Gly), developed by Vladimir Khavinson based on the bovine pineal extract Epithalamin. The signature finding is that Epitalon induced telomerase activity and elongated telomeres in cultured human fetal fibroblasts (Khavinson et al. Bull Exp Biol Med 2003).
That is a striking result, but it is cell culture. Animal work shows lifespan extension in some rodent strains. Human clinical data is mostly from Russian gerontology programs that are difficult to evaluate against Western standards. The proposed clinical use for the FDA review is still being clarified in the docket.
For Epitalon, the PCAC question is more fundamental than for the others: is "anti-aging" a clinical indication the FDA can credibly regulate, or is it a marketing frame for which no compounding pharmacy should be making bulk-drug-strength claims?
What the table doesn't show
The proposed uses are doing more work than they appear to. A peptide on the 503A list can only be compounded for the use the FDA specifies in the final rule. That means:
- A wound-healing nomination for TB-500 does not authorize compounding it for soft-tissue sports injuries
- A UC nomination for BPC-157 does not authorize gut-recovery use in IBS or general tendon repair
- A cerebral-ischemia nomination for Semax does not authorize cognitive enhancement in healthy users
Most of the off-label demand for these peptides falls outside the proposed uses. PCAC approval for the narrow indication does not legalize the broader research-use market.
What PCAC outcomes would actually mean
Three rough scenarios:
- PCAC recommends inclusion, FDA agrees, formal rule issued. Compounding for the specified use becomes legal and stable. Off-label compounding remains in a grey zone but with more legal risk because the on-label use is now defined.
- PCAC recommends against, FDA agrees. The peptide returns to a posture similar to Category 2: pharmacies will mostly stop compounding, and the "research peptide" market becomes the dominant supply channel.
- PCAC split, or FDA disagrees with PCAC. Status quo: compounders must make their own risk decisions, state pharmacy boards interpret the rules differently, and the legal picture stays unsettled.
Even scenario 1 is slow. Notice-and-comment rulemaking after a positive PCAC vote can take 12 to 24 months before a final rule is published.
Tip: If you want your view on the record, the docket (FDA-2025-N-6895) accepts written comments through July 22, 2026. Comments received by July 9 are sent to the committee in advance; later comments are considered by FDA but not by the committee at the meeting.
What changes for researchers and pharmacies right now
Practically nothing changes between today and the meeting. The April reclassification took the seven peptides off the Category 2 "significant safety risks" list, which removed an explicit FDA enforcement signal. It did not authorize compounding. Bulks-list inclusion is still the operative legal step.
For oral peptide alternatives that do not depend on injectable compounding, options like MK-677 sit in their own regulatory bucket and are not part of this PCAC review.
For research-use sourcing of compounds in this review (BPC-157, TB-500, KPV, MOTS-c, DSIP, Semax, Epitalon), the supply chain has historically run through research-chemical vendors rather than compounding pharmacies. That is independent of FDA compounding decisions, but the reputational signal from a PCAC outcome can shift physician and clinic willingness to engage with these compounds.
If you are planning to start a protocol around any of these compounds, work backward from the proposed use and the published trial population, not from forum dosing folklore. The BPC-157 colitis evidence does not support the same dose or duration that BPC-157 tendon-recovery forum culture has settled on. The KPV anti-inflammation evidence is mostly oral, not injectable.
For deep-dive guides, see the BPC-157 dosing protocol, the BPC-157 vs TB-500 comparison, and the Reconstitution Calculator.
How Peptides:Enhanced will track this
We will publish updates after the July 23 vote on Day 1 and after the July 24 vote on Day 2, including:
- The final PCAC recommendation by compound
- Specific evidence each panel raised
- The proposed-use language as voted on
- Any sponsor presentation details that emerged
Anything published in the docket between now and July 22 also matters. We will summarize the major comments separately and link them.
For trusted research-use sourcing while the regulatory picture clarifies, Ascension Peptides offers injectables (BPC-157, TB-500, MOTS-c) with 50% off using code ENHANCED, and Limitless Biotech covers oral KPV and other oral peptides with code ENHANCED.
Bottom line by compound
Bottom line: TB-500 has the strongest human-trial case for its proposed wound-healing use. BPC-157 has the deepest preclinical literature but a thin human file. KPV and MOTS-c rest almost entirely on animal work. DSIP and Semax depend on small or non-Western trials. Epitalon's telomerase claim is striking in cell culture but has minimal Western clinical evidence. All seven face the same procedural hurdle even if PCAC says yes: rulemaking still has to happen before anything is officially on the 503A list.
This article is for educational and research purposes only. None of the seven peptides discussed here is FDA-approved for any indication. The clinical uses listed are FDA-published proposed uses for a regulatory advisory meeting; they are not endorsements, approvals, or claims of efficacy. Information about the July 2026 PCAC meeting reflects the public docket FDA-2025-N-6895 and FDA's published meeting agenda as of May 2026 and may change before the meeting. Doses, routes, and durations referenced are described as published in the cited literature; they are not protocols for individual use. Compounding regulations vary by state pharmacy board even where federal rules are unsettled. Consult a qualified clinician for any individual medical question.