At a glance
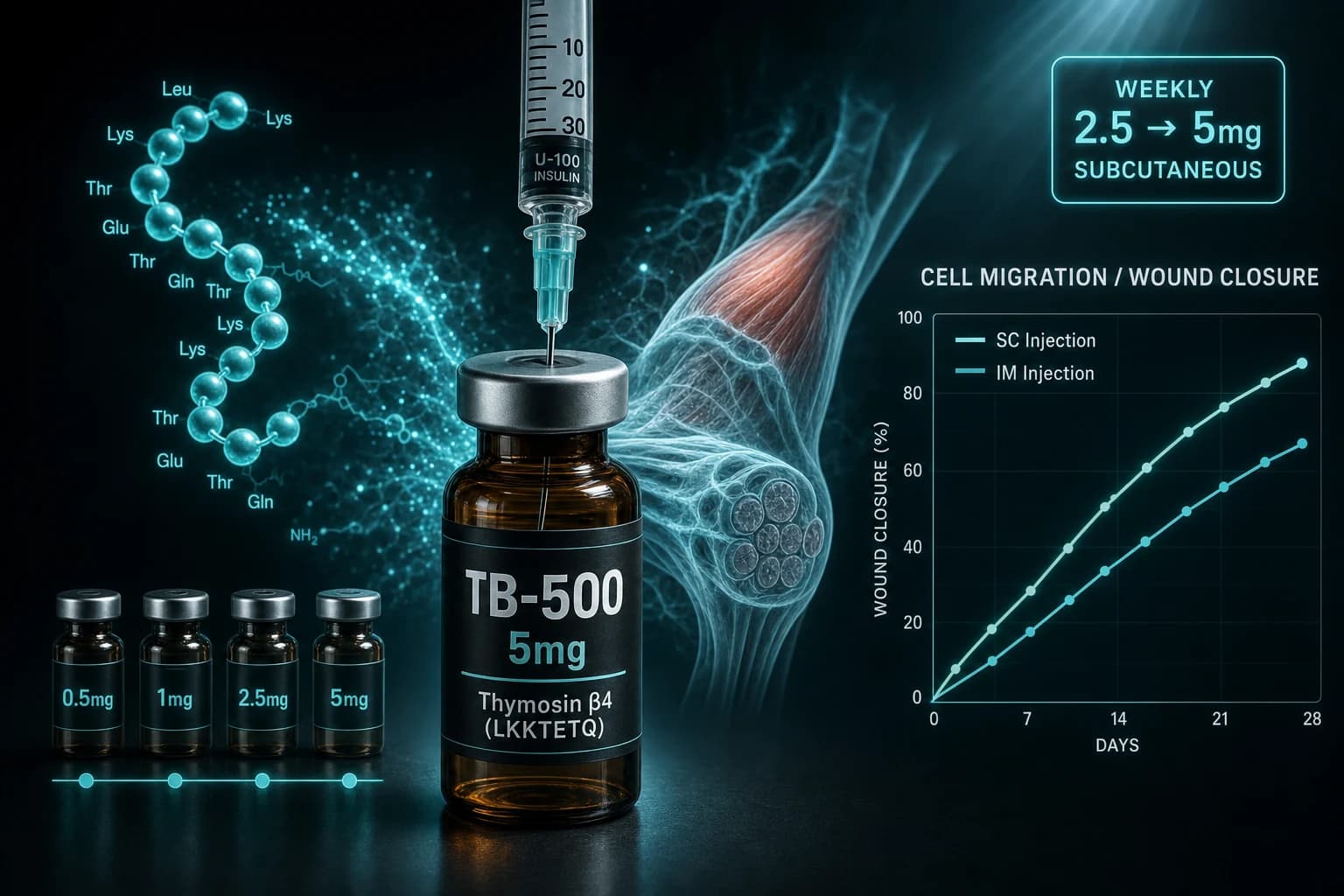
- Standard research protocol: 2.5 mg twice weekly loading for 4-6 weeks, then 2.5 mg weekly maintenance
- TB-500 is the synthetic LKKTETQ active region of Thymosin β4, not the full 43-residue protein
- Reconstitute a 5 mg vial with 2 mL bacteriostatic water for 0.5 mL = 1.25 mg per draw
- Subcutaneous and intramuscular routes both appear in the animal literature; SC is the practical default
- Most-cited mechanism is G-actin sequestration plus angiogenesis and cell migration; human RCT data is thin
TB-500 Dosage Guide: Loading, Maintenance, and Reconstitution Math
If you are looking up a TB-500 dose, you are most likely already running BPC-157 or the Wolverine Stack and trying to layer in actin-mediated tissue repair on top of angiogenic healing. The dosing question is the practical one: how much, how often, by what route, for how many weeks.
This guide covers the standard research-grade protocol used across published animal models and forum-documented human research, the reconstitution math for the typical 5 mg vial, the loading-versus-maintenance split that almost every user runs, and what the actual peer-reviewed literature says about TB-500 in tissue repair.
The standard research protocol
Across published preclinical work and forum-documented research-use protocols, the convergent dose is 2.5 mg twice weekly during loading, dropped to 2.5 mg once weekly for maintenance.
| Phase | Dose per injection | Frequency | Duration |
|---|---|---|---|
| Loading | 2.5 mg | Twice weekly (Mon/Thu) | 4 to 6 weeks |
| Maintenance | 2.5 mg | Once weekly | 4 to 8 weeks |
| Layered with BPC-157 | 2.5 mg TB-500 + 250-500 mcg BPC-157 daily | TB-500 weekly, BPC-157 daily | Run together for 4 weeks |
The loading phase loads systemic Thymosin β4 levels above background to activate the cell-migration and angiogenesis programs the peptide is best known for. The maintenance phase keeps levels elevated without pushing total weekly exposure higher than the literature has tested.
Two reasons most protocols cap loading at 6 weeks. First, the published wound-healing studies that drive the dosing rationale are mostly 14-to-28-day animal models (Malinda et al., FASEB J, 1999; Philp et al., FASEB J, 2003). Extrapolating beyond that window into human research is speculative. Second, anti-Tβ4 antibodies have been observed in long-duration animal exposure work, and capping loading bounds that risk.
What TB-500 actually is
A common point of confusion: TB-500 is not the full Thymosin β4 protein. It is a synthetic 17-amino-acid fragment marketed as containing the LKKTETQ active region of Tβ4, the part responsible for the actin-binding and cell-migration effects.
The full Thymosin β4 protein is a 43-residue peptide naturally present in nearly every cell type, where it is the major sequester of monomeric G-actin in the cytoplasm. TB-500 mimics the active region without the full molecular weight, which is why it ships as a research-grade synthetic peptide rather than a recombinant protein.
For mechanistic depth, Goldstein et al., Annals NY Acad Sci, 2010 and Sosne et al., Expert Opin Biol Ther, 2012 summarize the Tβ4 literature on cell migration, angiogenesis, and tissue repair across multiple injury models including dermal wounds, corneal burns, and cardiac repair.
Why dosing splits into loading and maintenance
Tβ4's mechanism front-loads work. The peptide binds G-actin, sequesters monomeric actin, and regulates the actin polymerization needed for cell migration. The cells you want to migrate (fibroblasts, endothelial cells, stem cells) need elevated Tβ4 levels during the active repair phase, not after.
For an acute injury, the active repair phase is roughly weeks 2 through 6 post-injury. Loading at 5 mg per week (2.5 mg × 2) front-loads exposure during that window. After week 6, the proliferative and remodeling phases of healing dominate, and Tβ4 levels can drop without losing the angiogenic gains already established.
Maintenance dosing exists for two reasons. First, weekly 2.5 mg keeps systemic Tβ4 above background while the remodeling phase plays out, in case there is residual benefit. Second, for chronic or recurrent injuries (chronic tendinopathy, repeat soft-tissue strains), running a longer maintenance cycle covers the protective benefit without escalating toward the upper end of what the literature has tested.
Reconstitution math for a 5 mg vial
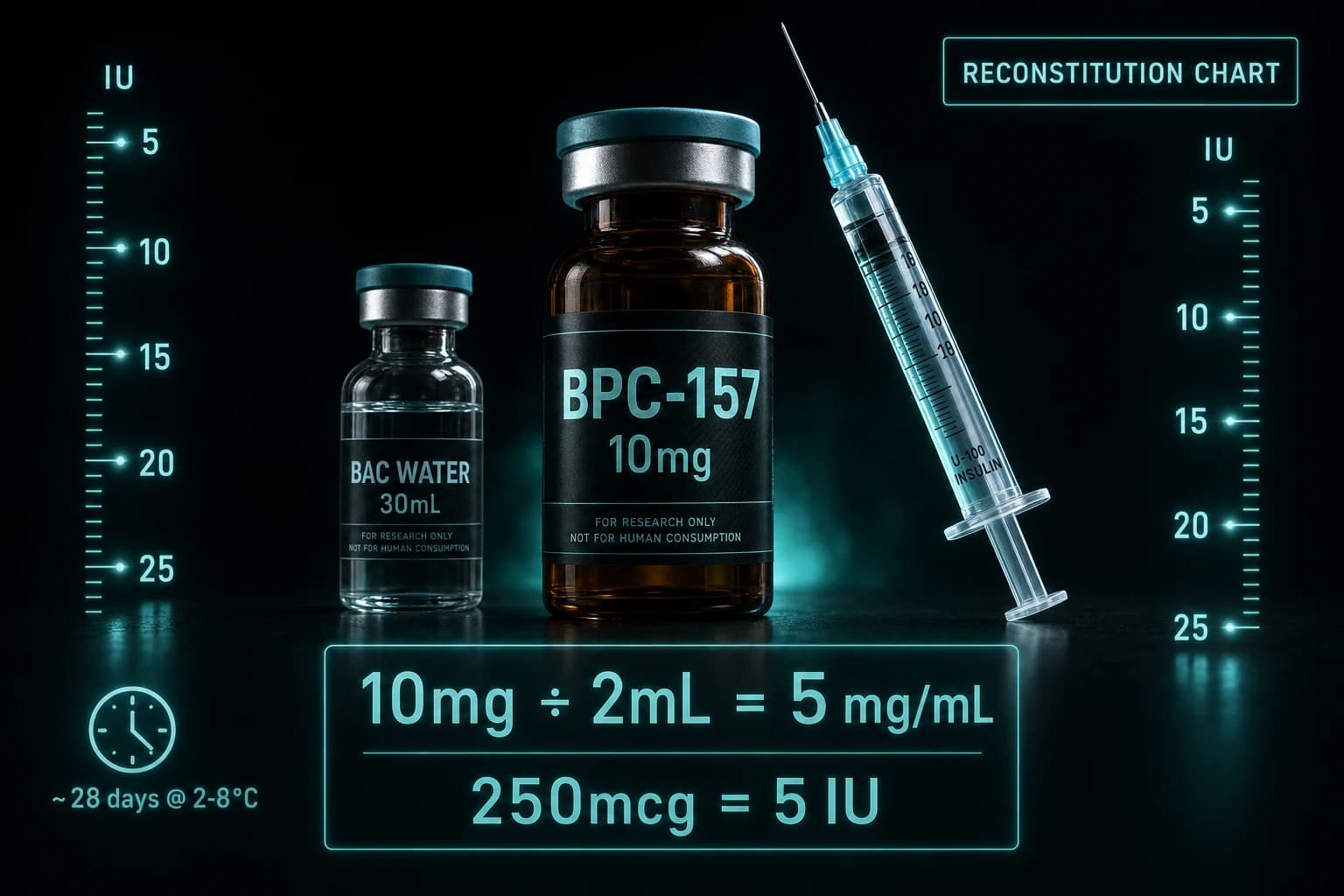
The most common vial size from research-grade vendors is 5 mg lyophilized powder. The reconstitution math determines how many milligrams sit in each syringe draw.
| Vial mass | Bacteriostatic water added | Concentration | 0.5 mL draw delivers |
|---|---|---|---|
| 5 mg | 1.0 mL | 5.0 mg/mL | 2.5 mg |
| 5 mg | 2.0 mL | 2.5 mg/mL | 1.25 mg |
| 5 mg | 2.5 mL | 2.0 mg/mL | 1.0 mg |
For the standard 2.5 mg dose, the cleanest reconstitution is 5 mg into 1 mL bacteriostatic water. A 0.5 mL draw on a U-100 insulin syringe (50 IU mark) delivers exactly 2.5 mg.
If you want finer titration (for example, splitting into 1.25 mg twice-weekly doses while testing tolerance), reconstitute with 2 mL. The same 0.5 mL draw now delivers 1.25 mg, and the vial yields four doses instead of two.
The reconstitution calculator handles arbitrary vial sizes and draw volumes if your vial is not 5 mg or your target dose differs.
Subcutaneous versus intramuscular
Both routes appear in the animal literature. The horse-doping study by Esposito et al., Drug Test Anal, 2012 characterized TB-500 pharmacokinetics in equine plasma after a single 10 mg IM dose. The corneal-injury work used topical drops. The dermal wound and tendon repair animal models typically used SC or IP injection.
For practical research dosing, subcutaneous is the default. Reasons:
- Pain and convenience. SC into abdominal fat is painless with a 31-gauge insulin needle. IM requires a longer needle and is more painful for weekly research dosing.
- Systemic distribution. TB-500 is a small peptide. SC injection achieves systemic distribution within hours, which is what you want for whole-body cell migration effects.
- Less local irritation. IM TB-500 has been reported to cause more localized soreness in forum-documented protocols.
If the research goal is localized injury (a specific tendinopathy or muscle strain), some researchers split the dose: half SC for systemic loading, half subcutaneous near the injury site. This is anecdotal and not supported by published comparative trials.
Stacking with BPC-157
The most common research stack pairs TB-500 with BPC-157. The two peptides target different healing mechanisms:
| Peptide | Primary mechanism | Local vs systemic | Standard research dose |
|---|---|---|---|
| TB-500 | G-actin sequestration, cell migration, angiogenesis | Systemic | 2.5 mg twice weekly |
| BPC-157 | Growth factor upregulation, VEGF, tendon-ligament repair | Local at injection site | 250-500 mcg daily |
The mechanistic rationale for the combination is that TB-500 prepares the systemic environment for cell migration and angiogenesis while BPC-157 drives local growth factor expression at the injury site. This is the same logic behind the pre-mixed Wolverine Stack, which combines both peptides in a single vial.
For a deeper mechanistic comparison of how the two peptides differ in cellular targets, the BPC-157 vs TB-500 deep dive covers the published evidence on each.
What the literature actually shows
Most TB-500 evidence is preclinical. The strongest published data comes from rodent and equine work, not human RCTs.
Dermal wound healing. Malinda et al., FASEB J, 1999 showed Thymosin β4 accelerated dermal healing of full-thickness punch wounds in normal rats. Subsequent work extended this to steroid-treated rats, diabetic mice, and aged mice. The effect was reproducible across labs.
Angiogenesis and hair follicle development. Philp et al., FASEB J, 2003 demonstrated Tβ4 promotes angiogenesis, wound healing, and hair follicle development in mouse models. The angiogenesis effect is a major part of the rationale for TB-500 in tendinopathy research, where impaired blood supply is a known healing bottleneck.
Corneal wound healing. Sosne et al., Exp Eye Res, 2002 showed topical Tβ4 promoted corneal wound healing and decreased inflammation following alkali injury. RegeneRx subsequently advanced Tβ4 ophthalmic formulations into human dry-eye trials.
Ligament repair. Xu et al., Knee Surg Sports Traumatol Arthrosc, 2013 showed Tβ4 enhanced healing of medial collateral ligament injury in rat. This is one of the more directly relevant studies for the musculoskeletal use case TB-500 is most often researched against.
Burn wounds. Banchaabouchi et al., 2014 reported Tβ4 improved burn wound healing in db/db diabetic mice via downregulation of receptor for advanced glycation end products.
What the literature does not have: large randomized controlled trials in humans for tendinopathy, muscle strain, or post-surgical recovery at TB-500 doses. The evidence base is animal-heavy, and any extrapolation to human research dosing carries that caveat.
Safety and what is known about adverse events
In rodent studies and the limited human trial work on full Thymosin β4 (the cardiac and dermal trials by RegeneRx), the safety profile has been generally favorable at the doses tested. Reported side effects in animal work include localized injection-site irritation and mild lethargy at the high end of dosing.
Two flags worth noting for research:
- Anti-Tβ4 antibodies. Long-duration exposure in animal work has produced anti-Tβ4 antibodies in some subjects. This is part of why most protocols cap loading at 4-6 weeks rather than running indefinitely.
- Theoretical angiogenesis concern. Tβ4 promotes angiogenesis. There is a theoretical concern that this could feed pre-existing angiogenesis-dependent pathology (notably tumor neovascularization). No clinical data supports this concern, but the mechanism is plausible enough that it appears in most TB-500 risk discussions.
WADA status
TB-500 is on the World Anti-Doping Agency prohibited list under S2 (Peptide Hormones, Growth Factors, Related Substances and Mimetics) at all times in and out of competition. Athletes in WADA-compliant testing pools should not run TB-500. The Esposito doping-control paper cited above exists specifically because regulatory bodies in equine sport have flagged TB-500 use.
Sourcing
For research-grade injectable TB-500 with public per-batch COAs, Ascension Peptides ships 5 mg vials with 50% off using code ENHANCED. The Wolverine Stack (pre-mixed BPC-157 + TB-500) is in the same catalog if the research protocol calls for both peptides in a single reconstitution.
For our deeper sourcing analysis, the best legit peptide vendors 2026 ranking covers vendor-by-vendor COA standards, and the where-to-buy BPC-157 with COAs guide is the closest analog for what to verify when buying a recovery peptide.
FAQ
What is the standard TB-500 research dose?
The convergent research-grade protocol is 2.5 mg twice weekly during a 4-to-6-week loading phase, dropped to 2.5 mg once weekly during maintenance. This is the dose that appears across most published animal-model adaptations and forum-documented human research protocols. Higher loading doses (5 mg twice weekly) appear in some protocols but lack additional published support.
Is TB-500 the same as Thymosin beta-4?
No. TB-500 is a synthetic 17-amino-acid fragment containing the LKKTETQ active region of Thymosin β4. The full Tβ4 protein is 43 residues. TB-500 mimics the actin-binding active region but is not the complete molecule. Most of the published mechanistic literature uses full-length Tβ4, and the assumption that the active fragment reproduces the same effects is mechanistically reasonable but not exhaustively proven.
How long should I run a TB-500 cycle?
Most research protocols cap loading at 4-6 weeks plus an optional 4-8 weeks of weekly maintenance. The published animal data are mostly in the 2-to-4-week range, so longer cycles extrapolate beyond what the literature has directly tested. Anti-Tβ4 antibodies have been documented in long-duration animal exposure, which is the practical reason most research protocols stop at 12 weeks total.
Can I run TB-500 and BPC-157 together?
Yes, this is the most-researched recovery peptide stack. The two peptides target different healing mechanisms: TB-500 drives systemic cell migration and angiogenesis, BPC-157 drives local growth factor expression. Standard combined research dosing is 2.5 mg TB-500 weekly plus 250-500 mcg BPC-157 daily, run together for 4-6 weeks. The pre-mixed Wolverine Stack ships both peptides in a single vial for convenience.
Subcutaneous or intramuscular for TB-500?
Subcutaneous is the practical default for systemic dosing. SC injection into abdominal fat with a 31-gauge insulin syringe is painless and reaches systemic circulation within hours. IM is sometimes used for localized injuries near the injection site, but the comparative evidence for local IM versus systemic SC at the same total dose does not clearly favor one approach.
Is TB-500 detectable in drug testing?
Yes. TB-500 is on the WADA prohibited list and equine drug-testing labs have published validated mass spectrometry methods for detection (Esposito et al., 2012). The detection window is at least several days post-injection. Athletes in WADA-compliant testing pools should not use TB-500.
Further reading
- BPC-157 vs TB-500: Recovery peptide comparison
- Wolverine Stack: BPC-157 + TB-500 protocol
- BPC-157 dosing protocol guide
- Where to buy BPC-157 with COAs
- Reconstitution Calculator
- TB-500 compound guide
This article is for educational and research purposes only. TB-500 is sold under research-use disclosures and is not approved by the FDA for any indication. None of the dosing protocols described should be interpreted as medical advice. WADA-tested athletes should not use TB-500. Consult a qualified clinician for individual medical questions.