At a glance
- 5-Amino-1MQ is a small molecule, not a peptide, that blocks NNMT and preserves cellular NAD+ and SAM
- Neelakantan 2018: 20 mg/kg/day reversed diet-induced obesity in mice with no change in food intake
- Neelakantan 2019: NNMT inhibition increased peak muscle torque ~70% in aged mice via stem cell reactivation
- Brachs 2019: genetic NNMT loss improved insulin sensitivity but did not change glucose tolerance
- Zero published human clinical trials as of May 2026; all efficacy data are rodent or cell culture
A research compound that does not look like a peptide
Most of the compounds people lump under "peptides" are actually peptides. 5-Amino-1MQ is not. It is a small synthetic quinolinium molecule that crosses cell membranes and binds the active site of nicotinamide N-methyltransferase, an enzyme that quietly drains NAD+ and methyl groups from fat and muscle when it is overexpressed.
That detail matters because the compound is sold and discussed in the same vendor channels as oral BPC-157 and oral MK-677, but the mechanism, the evidence base, and the safety profile sit in a different category. The honest version of the 5-Amino-1MQ story is that the rodent data are interesting and consistent, the human data do not exist yet, and the marketing is years ahead of the trials.
This review walks through what each of the key papers actually showed, where the evidence is strong, where it is weak, and where the gap between mouse and human research is being papered over with confident-sounding marketing.
What NNMT does and why anyone targets it
Nicotinamide N-methyltransferase is an enzyme that takes nicotinamide (a precursor your cells use to make NAD+) and methylates it using S-adenosylmethionine (SAM, the cell's main methyl donor) to produce 1-methylnicotinamide (MNA) and S-adenosylhomocysteine. When NNMT is overexpressed, two things happen at once. NAD+ precursor gets siphoned off, so cellular NAD+ falls. And SAM gets consumed, so methylation reactions across DNA, histones, and other substrates shift.
In healthy young tissue, baseline NNMT activity is low and the system is fine. In two specific conditions, baseline activity climbs sharply.
The first is obese white adipose tissue. The second is aged skeletal muscle. Both are tissues where falling NAD+ and altered methylation track closely with the metabolic dysfunction researchers most want to reverse.
Note: NNMT inhibition does not raise NAD+ everywhere in the body. It mostly affects tissues where NNMT is upregulated, which is why preclinical effects show up most clearly in obese fat and aged muscle rather than in young healthy animals.
The Kraus 2014 paper that started it
The case for NNMT as an obesity target rests heavily on Kraus et al. (2014), published in Nature. The team used antisense oligonucleotides to knock down NNMT specifically in fat tissue of mice on a high-fat diet.
Knockdown mice gained less weight than controls despite eating the same amount of food. Their adipose SAM and NAD+ levels both rose. They had increased polyamine flux (urinary diacetylspermine went up), elevated fat-cell oxygen consumption, and improved insulin sensitivity. The authors framed NNMT as a "novel regulator of histone methylation, polyamine flux, and NAD+-dependent SIRT1 signalling," a unique target for obesity and type 2 diabetes.
This paper did not use 5-Amino-1MQ. It used genetic knockdown. But it set up the rationale for everything that followed: if you can block NNMT pharmacologically without hitting other methyltransferases, you might get the same metabolic shift in a pill.
How 5-Amino-1MQ entered the picture
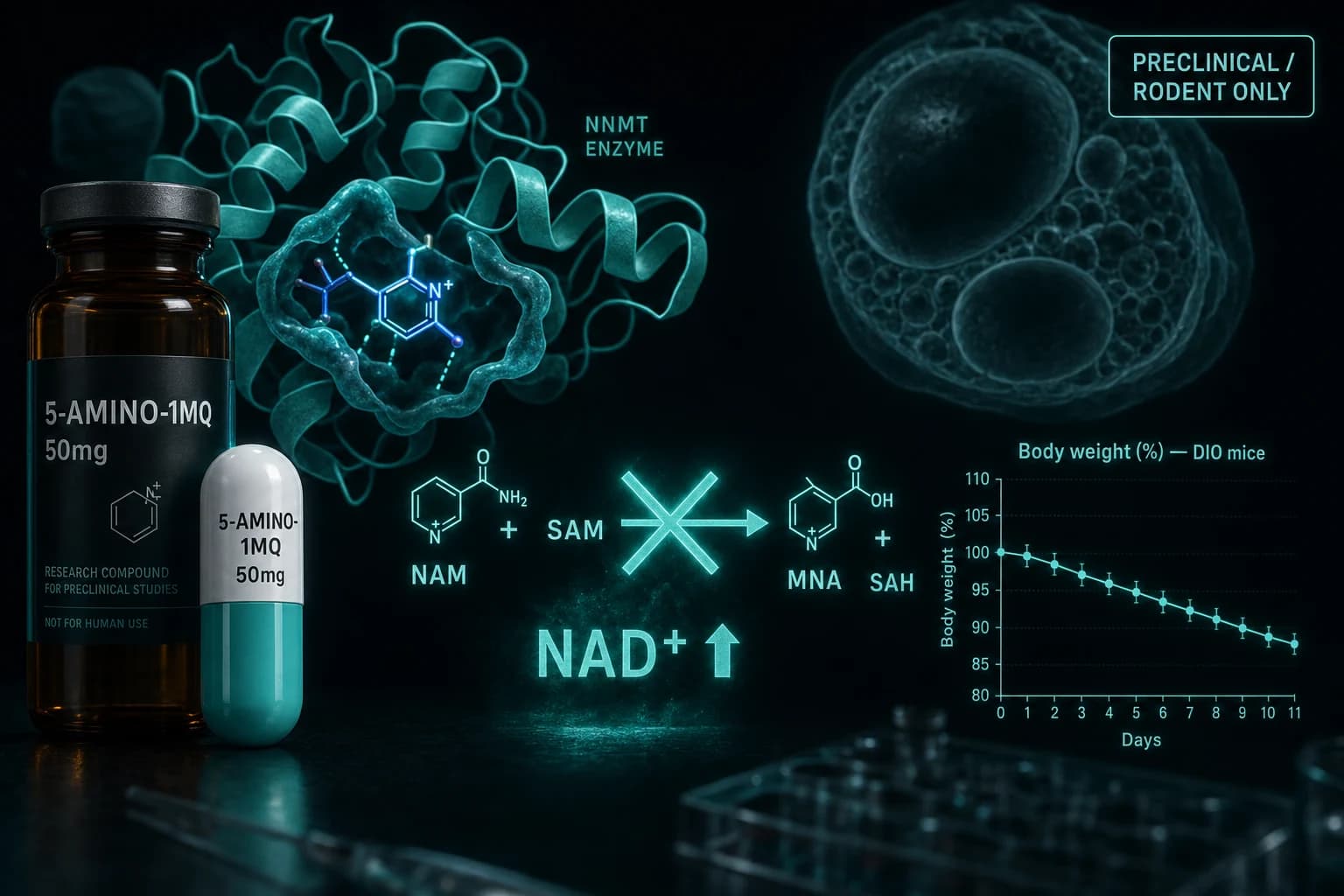
The defining 5-Amino-1MQ paper is Neelakantan et al. (2018) in Biochemical Pharmacology. The team screened quinolinium analogs for NNMT-selective inhibition, characterized binding kinetics, and confirmed cell membrane permeability. 5-Amino-1MQ emerged as the lead compound: selective for NNMT over related methyltransferases, IC50 in the low micromolar range, and able to reach intracellular NNMT in adipocytes.
In cell culture, 5-Amino-1MQ raised intracellular NAD+, lowered MNA, and suppressed lipogenesis in differentiated adipocytes. Diet-induced obese (DIO) mice received subcutaneous 5-Amino-1MQ at roughly 20 mg/kg/day for 11 days. Body weight, white adipose mass, and adipocyte size all dropped versus vehicle controls. Food intake did not change.
That last detail is the single most-cited finding in the consumer marketing of 5-Amino-1MQ, and it is the reason the compound gets framed as an alternative to GLP-1 weight-loss drugs. Ozempic and Mounjaro work largely by suppressing appetite. NNMT inhibition in this study did not. The fat loss came from a metabolic shift, not from eating less.
Note: "Did not significantly affect food intake" in an 11-day DIO mouse study is not the same thing as "preserves appetite over a year of human use." Read the marketing copy with that distinction in mind.
Evidence hierarchy at a glance
| Tier | Study | Model | What was shown |
|---|---|---|---|
| Foundational | Kraus 2014, Nature | Adipose-specific NNMT knockdown in DIO mice | Weight protection, increased NAD+ and SAM, improved insulin sensitivity |
| Lead compound | Neelakantan 2018, Biochem Pharmacol | DIO mice, 11 days, ~20 mg/kg/day SC | Reduced body weight and white adipose mass; food intake unchanged |
| Muscle stem cells | Neelakantan 2019, Biochem Pharmacol | Aged mice, NNMT inhibitor | Reactivated senescent satellite cells; ~70% peak torque increase versus controls after injury |
| Genetic dissection | Brachs 2019, Diabetes | NNMT knockout mice on high-fat diet | Improved insulin sensitivity; no change in glucose tolerance |
| Combination | Dimet-Wiley 2022, Sci Rep | DIO mice, NNMT inhibitor + low-fat diet | Greater weight and adiposity loss than either alone, with a distinct gut microbiome shift |
| Human | None published | n/a | No completed RCTs as of May 2026 |
The pattern across these papers is consistent at the mechanism level. Block or remove NNMT, raise tissue NAD+ and SAM, get downstream effects on adiposity, insulin sensitivity, or muscle quality. The pattern is also entirely preclinical. There is no human Phase 1 dose-finding study, no human pharmacokinetic publication on 5-Amino-1MQ, and no published placebo-controlled trial in any indication.
The muscle stem cell finding that changed the story
The 2018 fat paper got the press, but the more interesting paper for healthspan researchers is Neelakantan et al. (2019), also in Biochemical Pharmacology. Aged skeletal muscle accumulates NNMT, loses NAD+, and shows reduced satellite cell (muscle stem cell) activity. The team treated aged mice with an NNMT inhibitor and measured stem cell behavior, fiber regeneration after injury, and contractile performance.
Treated mice had increased satellite cell proliferation, larger regenerating fibers after injury, and roughly a 70% increase in peak isometric torque compared with vehicle controls. The authors argued that NNMT inhibition reactivates senescent muscle stem cells by restoring the local NAD+ environment, with downstream effects on SIRT1 and the regenerative program.
This is the part of the literature that connects 5-Amino-1MQ to the broader sarcopenia and longevity conversation. It is also the part most often overstated. The 70% number applies to peak torque after a specific injury model in aged mice. It does not mean a 70% strength increase in middle-aged humans, and the authors do not claim that. Forum copy that strips the model and the context is the problem, not the paper.
Where the NNMT story breaks down
The most useful corrective in the literature is Brachs et al. (2019) in Diabetes. The team used full-body NNMT knockout mice, not adipose-specific antisense knockdown, and ran them through a high-fat diet challenge.
Whole-body NNMT knockout mice did improve their basal metabolic parameters and insulin sensitivity, consistent with the Kraus model. But they did not show improved oral glucose tolerance, and they did not show the same magnitude of weight protection seen with adipose-specific knockdown. The authors concluded that the metabolic benefits of NNMT loss are real but are tissue-specific and incomplete.
That is the most important nuance for anyone considering 5-Amino-1MQ as a "metabolic health" compound. Genetic NNMT loss in a controlled mouse model gives a partial metabolic benefit, not a wholesale one. Pharmacologic inhibition with a small molecule for short periods in DIO mice has not been replicated in long-duration human studies of any kind.
Bottom line: The mechanism is real. The rodent metabolic benefits are reproducible. The size of the human effect is unknown. Anyone selling 5-Amino-1MQ as a Phase-3-grade weight loss tool is years ahead of the data.
Dosing, half-life, and the absence of human pharmacokinetics
Vendor websites typically describe 5-Amino-1MQ in 50 mg or 150 mg oral capsules taken once or twice daily. None of those protocols come from a published human study. They are extrapolations from the 20 mg/kg/day mouse data scaled by allometric body-surface conversion or just by analogy.
There is no published human Tmax, no human half-life, no human Cmax, no published bioavailability for the oral form, and no peer-reviewed dose-finding work. The compound is also not listed on any FDA-approved drug label, which means the standard tools you would use to compare oral semaglutide (Rybelsus) or oral orforglipron do not exist for 5-Amino-1MQ.
If you are weighing this compound against established options for fat loss, the right comparison is not "5-Amino-1MQ versus tirzepatide" but rather "preclinical compound with no human PK data versus Phase 3 drugs with thousands of patient-years of evidence." That framing makes the choice clearer.
How NNMT inhibition fits among adjacent options
| Goal | Compound class | Evidence depth in humans | Realistic 2026 expectation |
|---|---|---|---|
| Significant weight loss | GLP-1 / GIP agonists (tirzepatide, retatrutide, oral orforglipron) | Phase 3 RCTs, n in thousands | 12 to 24% body weight reduction reported in trials |
| Visceral fat in HIV lipodystrophy | Tesamorelin | Phase 3, FDA-approved indication | Targeted visceral adipose reduction in defined population |
| Mitochondrial / exercise mimetic | MOTS-c | Animal and small human pilots | Mechanistic interest, not weight-loss tool |
| NAD+ pathway support | NR, NMN, NAD+ injection | Mostly small human studies | Modest, varied effects on energy and aging biomarkers |
| NNMT inhibition | 5-Amino-1MQ | Rodent only | Preclinical interest; treat as an unproven research compound |
The honest read of this table is that 5-Amino-1MQ does not directly compete with GLP-1 receptor agonists on weight loss. It belongs in the NAD+ pathway and longevity-research bucket, alongside other compounds where the mechanism is intriguing and the human evidence is thin. People stack it with GLP-1s hoping the muscle preservation hint from Neelakantan 2019 transfers, but that hope is mechanistic, not clinical.
What is missing from the file
A short list of what the literature does not yet have for 5-Amino-1MQ:
- A human Phase 1 PK and safety study
- Any human RCT in any indication
- Long-duration rodent toxicology beyond a few weeks
- Body composition data with DEXA or MRI in any species at relevant doses
- Direct head-to-head comparisons with GLP-1 agonists or with NMN/NR in matched models
- Published data on combination with GH secretagogues or GLP-1s in either rodents or humans
Until at least the first two are published, "5-Amino-1MQ for fat loss" is a research hypothesis, not a clinical option. That does not make the compound uninteresting. It makes the marketing claims premature.
Practical context for buyers
5-Amino-1MQ is sold under the same research-chemical framework as most early-stage compounds discussed on this site. It is not FDA-approved, the 503A compounding discussion does not include it, and oral capsules sold by research vendors are not regulated as drugs.
For people who do choose to source it for personal research, the relevant axis is purity, third-party testing, and route. Limitless Biotech covers oral 5-Amino-1MQ capsules and other oral compounds with code ENHANCED. For injectable adjuncts often discussed in the same metabolic-health context, like tesamorelin or MOTS-c, Ascension Peptides offers research-grade vials with 50% off using code ENHANCED, plus a reconstitution calculator for accurate dosing math.
The general rule that applies to every compound in this category: the strength of the marketing copy is inversely correlated with the strength of the evidence. Fact-check vendor claims against the actual papers, not against rephrased versions of those papers.
Warning: No published study supports any specific 5-Amino-1MQ dose for human use. Doses in the literature are mouse doses, often at body-surface-area equivalents that do not translate cleanly. Anyone using this compound is running an n=1 experiment without a published safety floor.
Bottom line
5-Amino-1MQ is a real research tool with a clear mechanism and a small, internally consistent rodent literature. The Kraus paper established NNMT as an obesity-relevant target. Neelakantan 2018 showed pharmacologic inhibition reproduces the fat-loss phenotype in DIO mice without changing food intake. Neelakantan 2019 connected the same target to muscle stem cell function in aging. Brachs 2019 added the necessary caveat that not every metabolic axis improves with NNMT loss.
What the literature does not have is a single human trial. Treat 5-Amino-1MQ the way you would treat any preclinical compound: interesting, worth tracking, not a substitute for evidence-based options like GLP-1 receptor agonists or established lifestyle interventions. If a Phase 1 trial publishes in the next few years, the conversation changes. Until then, this is research-grade interest, not a clinical recommendation.
For deeper reading on the adjacent space, the GLP-1 muscle preservation review and the tesamorelin visceral fat guide cover the human-trial side of metabolic peptides. The oral versus injectable bioavailability guide explains why oral compounds in this category face an uphill battle on PK alone.
This article is for educational and research purposes only. 5-Amino-1MQ is not FDA-approved for any indication and has no published human clinical trials as of May 2026. Doses, durations, and route descriptions in this article reflect what is reported in the cited preclinical literature; they are not protocols for human use. Nothing here is medical advice. Consult a qualified clinician for individual medical questions.