At a glance
- Rybelsus is oral semaglutide with SNAC permeation enhancer; approved at 3, 7, 14 mg once-daily for type 2 diabetes
- Orforglipron is a small-molecule oral GLP-1 (not a peptide); Phase 3 ATTAIN program tested doses up to 36 mg with about 12% weight loss
- Rybelsus requires strict fasted dosing with 4 oz water and a 30 minute food/drink wait; orforglipron has no food restrictions
- Injectable semaglutide produces more weight loss than either oral option; oral routes trade efficacy for convenience
- Compounded oral peptide capsules are a separate category from approved orals and use different absorption strategies
The oral GLP-1 conversation in 2026 is no longer just about Rybelsus. Orforglipron is filed with the FDA and the ATTAIN Phase 3 program produced about 12% weight loss at the 36 mg dose. The class is now a real choice between two structurally different oral options, plus a separate market of research-grade oral peptide capsules.
This article covers what each option actually is at the molecule level, how they dose, what the trial efficacy looks like, and the practical food-rule and bioavailability differences that determine which oral GLP-1 fits which use case.
Two different molecules at the oral route
The first thing to get right: Rybelsus and orforglipron are not the same kind of compound.
Rybelsus is semaglutide, the same peptide as Ozempic and Wegovy, formulated with the absorption enhancer SNAC (sodium N-(8-[2-hydroxybenzoyl] amino) caprylate). SNAC transiently raises gastric pH at the absorption site and protects semaglutide from proteolytic degradation long enough to cross the gastric mucosa. This is what makes a peptide bioavailable orally at all. Rybelsus is approved for type 2 diabetes at 3, 7, and 14 mg once-daily.
Orforglipron is a small-molecule GLP-1 receptor agonist developed by Lilly. It is not a peptide. It is an orally bioavailable small molecule that does not require absorption enhancement. The dose ladder runs to 36 mg once-daily, and the Phase 3 ATTAIN program covers both obesity and type 2 diabetes indications.
The mechanism downstream (GLP-1 receptor activation in pancreatic beta cells, brain satiety circuits, slowed gastric emptying) is the same. The molecule entering the body is structurally distinct, and that distinction drives everything from food rules to side effects to head-to-head efficacy.
For more on the orforglipron Phase 3 evidence and the small-molecule case, see the orforglipron Phase 3 evidence article.
Rybelsus: oral semaglutide with SNAC
Rybelsus was approved in 2019 for type 2 diabetes based on the PIONEER Phase 3 program.
| Trial | Population | Dose | Result | Reference |
|---|---|---|---|---|
| PIONEER 1 | T2D monotherapy | 3, 7, 14 mg | -1.4% A1C, -2.6 kg at 14 mg | Aroda et al., 2019 |
| PIONEER 4 | T2D vs liraglutide | 14 mg | Non-inferior A1C, superior weight loss vs liraglutide | Pratley et al., Lancet, 2019 |
| PIONEER 6 | T2D CV outcomes | 14 mg | Non-inferior CV safety | Husain et al., NEJM, 2019 |
Rybelsus is approved at 3 mg starter for 30 days, then escalates to 7 mg. If additional A1C reduction is needed, the dose increases to 14 mg after another 30 days. The 14 mg dose is the maintenance for most patients.
Mean weight loss at 14 mg in the diabetes population was approximately 2.6 kg at 26 weeks. This is meaningful for diabetes but well below what injectable semaglutide produces at higher doses for obesity. As of May 2026, Rybelsus is not approved for obesity at a higher dose, although a 25 mg oral semaglutide for obesity was investigated in earlier trials and reportedly produced approximately 17% weight loss at 68 weeks in obese non-diabetics.
The food rules that actually matter
Rybelsus has the most restrictive dosing protocol of any approved GLP-1. The label specifies:
- Take the tablet first thing in the morning, in the fasted state.
- Swallow with no more than 4 ounces (118 mL) of plain water.
- Wait at least 30 minutes before any food, drink, or other oral medication.
These rules are not optional. Bioavailability of oral semaglutide depends on the SNAC enhancer being able to act on the gastric mucosa without dilution or competitive absorption. A patient who takes Rybelsus with coffee or breakfast can reduce systemic exposure substantially. The PIONEER trial protocols enforced the food rules and the published efficacy data assumes adherence.
Patient adherence to the 30-minute fasted window is the main practical reason Rybelsus has not displaced injectable semaglutide in clinical practice despite being orally bioavailable. The convenience of an oral tablet evaporates if the patient has to wake up 30 minutes earlier than normal to take it correctly.
Orforglipron: small-molecule oral with no food rules
Orforglipron is structurally distinct from Rybelsus. As a small molecule, it does not require permeation enhancement and is absorbed through standard GI absorption pathways. The label, when finalized, is expected to have no food restrictions, which is the major practical advantage over Rybelsus.
The ATTAIN Phase 3 program tested doses up to 36 mg once daily in both obesity and type 2 diabetes populations.
| Trial | Population | Top dose | Top-dose weight loss | Duration |
|---|---|---|---|---|
| ATTAIN-1 | Obesity | 36 mg | ~12.4% | 72 wk |
| ATTAIN-2 | T2D | 36 mg | ~9.6% | 72 wk |
| ACHIEVE-1 | T2D add-on | 36 mg | ~7.9% A1C reduction relative to baseline | 52 wk |
| ACHIEVE-3 | T2D vs sitagliptin | 36 mg | superior A1C and weight loss | 52 wk |
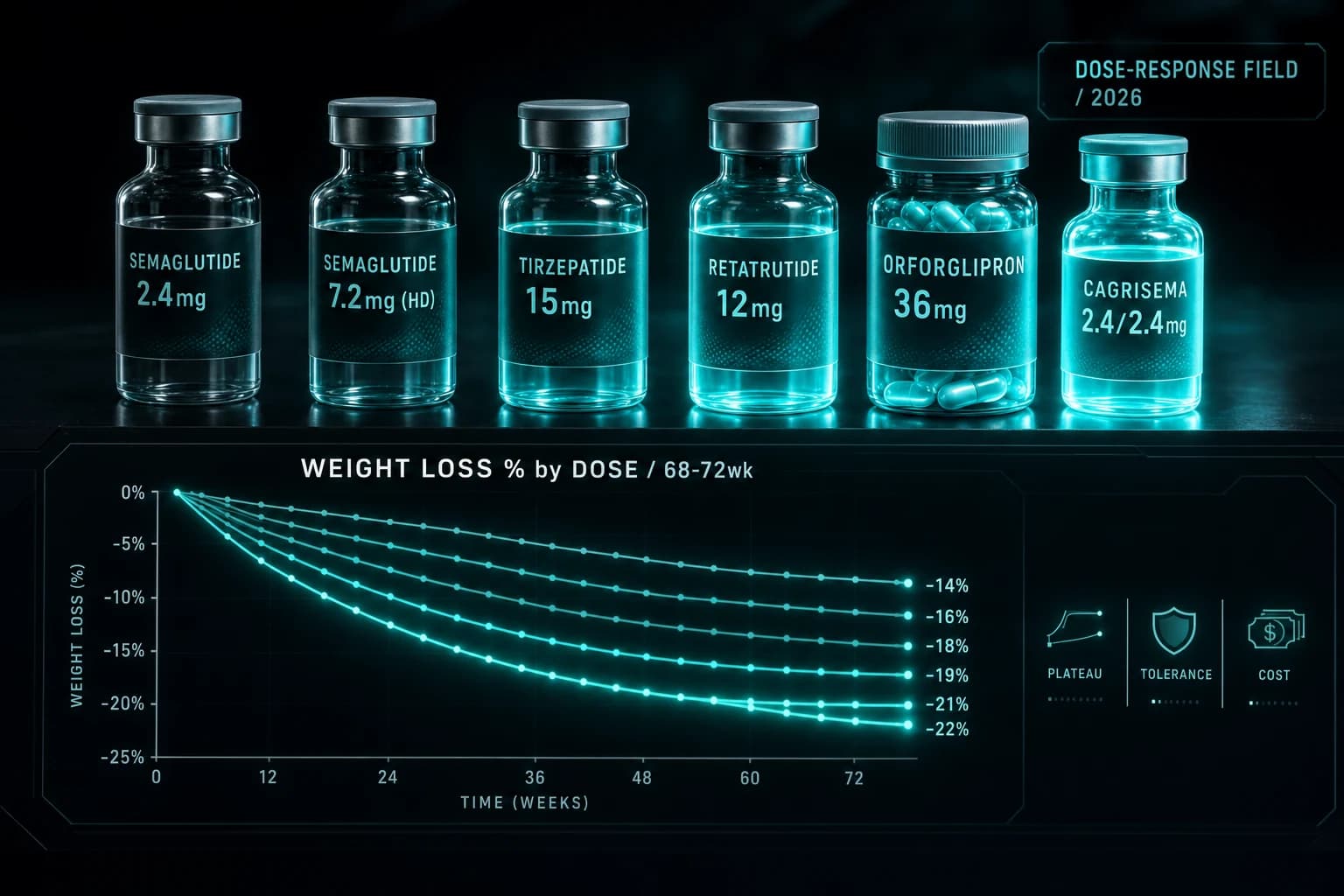
The orforglipron 36 mg result at approximately 12% weight loss in obesity is the most-marketed comparison point against injectable GLP-1s. Direct comparisons:
- Orforglipron 36 mg: ~12% at 72 wk (oral, no food rules)
- Rybelsus 14 mg: ~2.6 kg at 26 wk (oral, strict food rules, T2D label)
- Injectable semaglutide 2.4 mg: ~15% at 68 wk (injection, no food rules)
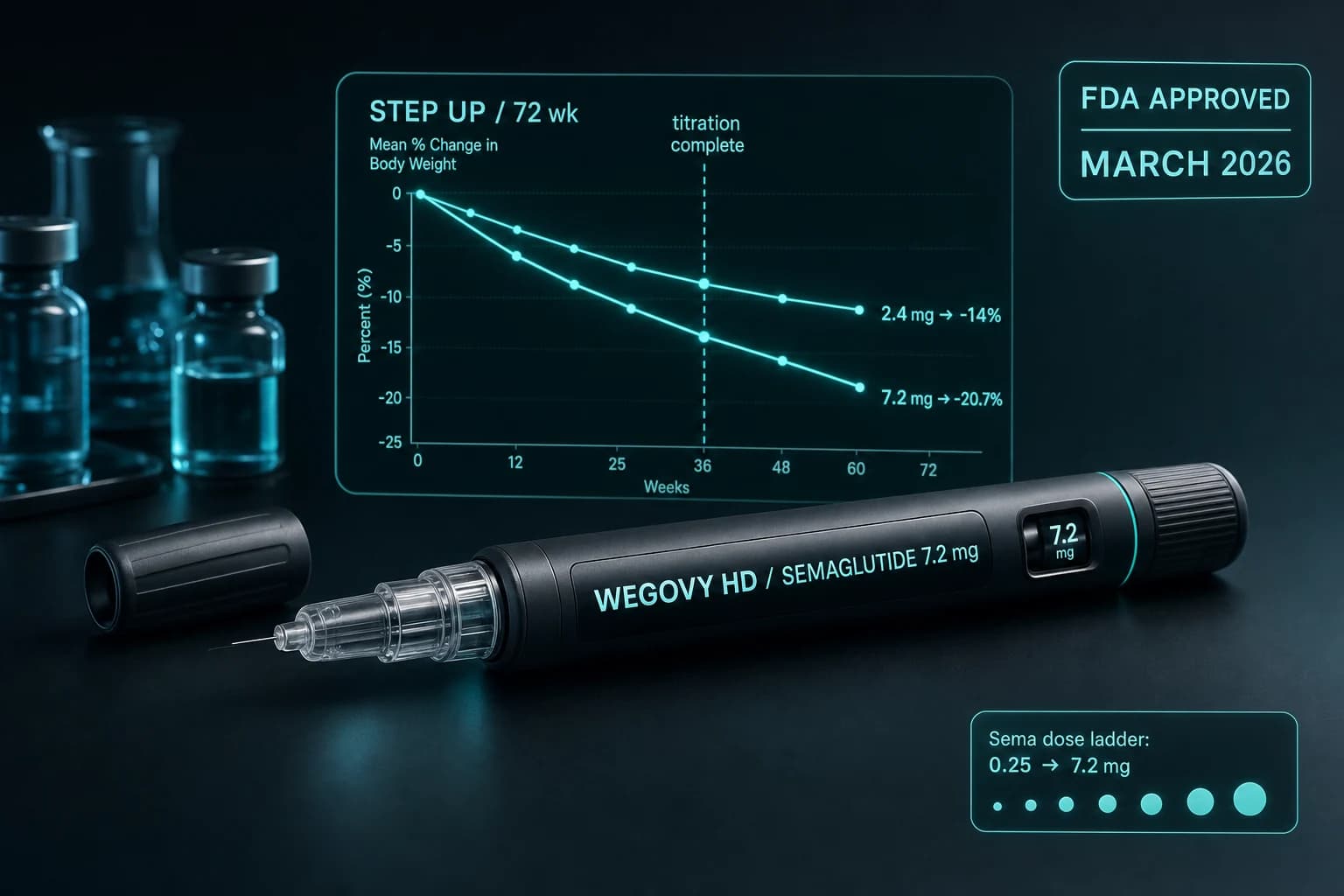
- Injectable semaglutide 7.2 mg (Wegovy HD): ~21% at 72 wk (injection, no food rules)
- Tirzepatide 15 mg: ~22% at 72 wk (injection, no food rules)
Orforglipron carved out a real position in the class as the no-food-rule oral GLP-1. The trade-off is that the weight loss is meaningfully below injectable semaglutide at the lowest approved obesity dose (2.4 mg).
For our complete dose-response coverage across the class, see the GLP-1 dosing comparison 2026.
Head-to-head: which oral GLP-1 for which use case
| Need | Best oral option | Why |
|---|---|---|
| Maximum oral weight loss | Orforglipron 36 mg | About 12% at 72 wk, no food rules |
| Type 2 diabetes with mild weight goal | Rybelsus 14 mg | Approved, established CV safety profile, 30 min food rule |
| Patient cannot tolerate food rules | Orforglipron | No restrictions |
| Patient already on injectable | Stay on injectable | Both orals produce less weight loss than injectable semaglutide |
| Cost-conscious oral | Rybelsus (typically lower than orforglipron) | Established generic competition timeline |
| Long-term oral monotherapy obesity | Orforglipron, once finalized | Designed for the obesity population |
The honest position: if maximum weight loss is the goal, neither oral matches injectable semaglutide. The oral route is for patients who will not or cannot inject, where the convenience trade-off is worth the efficacy gap. Within the oral category, orforglipron is the higher-efficacy and lower-restriction option, but it is not yet finalized as an obesity-indicated label.
Bottom line: For most weight-loss-focused patients, injectable semaglutide produces meaningfully more weight loss than either oral option. Choose oral when injection is a hard barrier. Within oral options, orforglipron beats Rybelsus on both efficacy and convenience (no food rules), but is still pre-approval as of May 2026.
For mechanism comparisons across the injectable class, see tirzepatide vs semaglutide 2026 and retatrutide vs tirzepatide vs semaglutide.
Side effect profiles compared
Both compounds share the GLP-1-class side effect signature: nausea, diarrhea, constipation, decreased appetite. The patterns at the oral route differ in nuance.
Rybelsus. Nausea is dose-dependent (more frequent at 14 mg than 3 mg). The food rule is partly responsible for the GI profile, because the gastric pH window where SNAC acts is also where nausea originates. Patients who take Rybelsus correctly tend to have nausea that resolves within 4-6 weeks of each dose step.
Orforglipron. GI side effects scaled with dose in the ATTAIN program. The 36 mg arm had higher discontinuation rates than 12 or 24 mg, with nausea and vomiting being the most common reasons. The absence of the SNAC permeation pathway means the GI profile is less dependent on timing.
Across both compounds, the side effect class is well-characterized and not surprising for clinicians familiar with injectable GLP-1s. Patients can expect a similar adaptation period at the oral route.
Compounded oral peptide capsules: a separate category
Research-grade oral peptide capsules are sold by vendors including Limitless Biotech under code ENHANCED. These are distinct from both Rybelsus and orforglipron in several ways:
-
Formulation strategy. Compounded oral peptides typically use lipid-based or absorption-enhancer formulations distinct from SNAC. The bioavailability achieved by any specific compounded product depends on the formulation, which varies by manufacturer.
-
Regulatory status. Compounded oral peptides are sold for research purposes, not as FDA-approved oral GLP-1 therapies. Compounded semaglutide and tirzepatide are most commonly available as injectable preparations because the oral route has not been validated outside of Rybelsus.
-
Other compounds at the oral route. Beyond GLP-1s, oral capsules of non-GLP-1 metabolic compounds (5-amino-1MQ, tesofensine, SLU-PP-332) are oriented toward different mechanisms entirely. For oral non-GLP-1 metabolic compounds with research-grade documentation, see the 5-amino-1MQ research review, tesofensine dosage guide 2026, and SLU-PP-332 dosage protocol.
The injectable vs oral peptides bioavailability guide covers the broader route-of-administration tradeoffs across the peptide research class.
What changes in 2026
Three factors are reshaping the oral GLP-1 conversation this year:
-
Orforglipron filing and expected approval. The Lilly filing in late 2025 means orforglipron is heading toward an approval decision in the obesity indication. If approved, this becomes the first true small-molecule oral GLP-1 for obesity. The current approved orals are oral semaglutide (Rybelsus, T2D only) and any future formulation expansions.
-
Wegovy HD set the new injectable ceiling. The Wegovy HD 7.2 mg STEP UP trial result of 20.7% at 72 weeks expanded the gap between oral and injectable semaglutide. Oral semaglutide at 14 mg now sits even further below the injectable ceiling than it did before HD.
-
Pipeline beyond GLP-1 monotherapy. The injectable class is moving toward multi-receptor agents (tirzepatide GIP/GLP-1, retatrutide GIP/GLP-1/glucagon, MariTide GIP-antagonist/GLP-1). The oral class is currently single-receptor (GLP-1 only). The mechanism gap between oral and injectable is widening, not closing.
For the broader pipeline coverage, see the survodutide GCG/GLP-1 dual agonist and mazdutide IBI362 Phase 3 evidence.
FAQ
Should I take Rybelsus or wait for orforglipron?
If you have type 2 diabetes and need an oral GLP-1 now, Rybelsus is approved and available. If your primary goal is obesity weight loss at the oral route, the answer is more nuanced. Orforglipron at 36 mg in Phase 3 produced about 12% weight loss versus Rybelsus at 14 mg producing approximately 2.6 kg in T2D patients. The two products are aimed at different populations and approval indications. Talk to a clinician about which approved label and dose fits your goal.
Why does Rybelsus have such strict food rules?
Rybelsus is oral semaglutide combined with SNAC, a permeation enhancer that creates a transient gastric absorption window. Food, fluid beyond 4 ounces of water, or other medications taken during that window dilute or compete with the SNAC mechanism and reduce systemic exposure. The 30-minute fasted wait protects the absorption window. The food rules are not optional, and adherence directly determines the efficacy you actually get from the dose.
Is orforglipron a peptide?
No. Orforglipron is an orally bioavailable small molecule GLP-1 receptor agonist. It is not a peptide. This is a structural distinction that drives the absence of food restrictions (because there is no peptide bioavailability problem to solve with permeation enhancement) and the higher mg dose range (because small molecules dose at different scales than peptides).
Can I get more weight loss from Rybelsus 25 mg than 14 mg?
The Rybelsus 25 mg dose was investigated in obesity trials and reportedly produced approximately 17% weight loss at 68 weeks in obese non-diabetics. As of May 2026, the 25 mg dose is not approved for obesity. The 14 mg dose is the current approved maintenance for T2D. Whether Novo Nordisk pursues a higher-dose Rybelsus obesity label is an ongoing pipeline question.
Can I combine Rybelsus and orforglipron?
Combining two GLP-1 receptor agonists is not a tested or supported protocol. The GLP-1 receptor saturation question, combined side effect burden, and overlapping pharmacology make this a poor research approach. Combination therapy in the class is structured around different receptor families (GLP-1 + amylin in CagriSema, GLP-1 + GIP in tirzepatide, GLP-1 + GIP + glucagon in retatrutide), not two agents on the same receptor.
How does oral GLP-1 compare to compounded oral peptide capsules?
Approved oral GLP-1s (Rybelsus, eventually orforglipron) have FDA-validated bioavailability and Phase 3 efficacy data. Compounded oral peptide capsules are research-grade products sold for research purposes, typically using different formulation strategies. They are not the same regulatory category, and the bioavailability of compounded oral products varies by manufacturer and formulation. For research-grade work, COA verification matters more for compounded oral products than for approved injectables.
Does orforglipron work for diabetes too?
Yes. The ATTAIN program covers both obesity and T2D indications. The ACHIEVE arms specifically tested orforglipron in type 2 diabetes against sitagliptin and demonstrated superior A1C reduction and weight loss. If approved, orforglipron will likely have both indications in the label.
Sourcing
For research-grade compounds outside the approved oral GLP-1 class, Ascension Peptides ships injectable peptides with 50% off using code ENHANCED including research-grade semaglutide and tirzepatide vials. Limitless Biotech ships oral metabolic capsules with code ENHANCED.
For broader sourcing analysis, see the best legit peptide vendors 2026 ranking. For format-aware purchasing context, see the injectable vs oral peptides bioavailability guide.
Further reading
- Orforglipron Phase 3 evidence: Lilly's oral GLP-1 in 2026
- Wegovy HD 7.2mg STEP UP trial guide
- GLP-1 dosing comparison 2026
- Tirzepatide vs Semaglutide 2026 head-to-head
- Injectable vs oral peptides: bioavailability guide
- Tesofensine dosage guide 2026
- 5-amino-1MQ NNMT inhibitor research review
- Best legit peptide vendors 2026
This article is for educational and research purposes only. Rybelsus (oral semaglutide) is FDA approved for type 2 diabetes. Orforglipron is filed with the FDA and is not yet approved as of May 2026. Compounded oral peptide capsules are sold for research purposes only. None of the content above constitutes medical advice. Consult a qualified clinician for individual medical questions about diabetes or weight management therapy.